The Chemical Context of Life
The Chemical Context of Life. Chapter 2. Matter. Matter consists of chemical elements in pure form and in combinations called compounds; living organisms are made of matter. Matter -- Anything that takes up space and has mass.


The Chemical Context of Life
E N D
Presentation Transcript
The Chemical Context of Life Chapter 2
Matter • Matter consists of chemical elements in pure form and in combinations called compounds; living organisms are made of matter. • Matter -- Anything that takes up space and has mass. • Element -- A substance that cannot be broken down into other substances by chemical reactions; all matter made of elements. • Life requires about 25 chemical elements • 96% of living matter is composed of C, O, H, N. • Most of remaining 4% is P, S, Ca, K. • Trace element -- required by organisms in extremely small quantities: Cu, Fe, I, etc.
Matter cont. • Compound -- Pure substances made of two or more elements combined in a fixed ratio. • Have characterisitics different than the elements that make them up (emergent property). • Na and Cl have very different properties from NaCl. • Difference between mass and weight: • Mass -- measure of the amount of matter an object contains; constant. • Weight -- measure of how strongly an object is pulled by earth's gravity; varies.
Atomic structure determines the behavior of an element • Atom -- Smallest possible unit of matter that retains the physical and chemical properties of its element. • Subatomic Particles • 1. Neutrons (no charge/neutral; found in nucleus; ~ 1 amu). • 2. Protons (+1 charge; found in nucleus; ~ 1 amu). • 3. Electrons (-1 charge; electron cloud; 1/2000 amu). • One amu approx equal to 1.7 x 10-24 g.
Atomic Number and Atomic Weight • Atomic number = Number of protons in an atom of a particular element. • All atoms of an element have the same atomic number. • In a neutral atom, # protons = # electrons. Mass number -- Number of protons and neutrons in an atom; not the same as an element's atomic weight.
Examples • 23Mg Mass number ?? Atomic number ?? • 12 • 23 12 • # of protons ?? # of electrons ?? # of neutrons ?? • 12 12 11 • 14C Mass number ?? Atomic number ?? • 6 • 14 6 • # of protons ?? # of electrons ?? # of neutrons ?? • 6 6 8
Isotopes • Isotopes -- Atoms of an element that have the same atomic number but different mass number; different number of neutrons. • Half-life -- Time for 50% of radioactive atoms in a sample to decay. • Biological applications of radioactive isotopes include: • 1. Dating geological strata and fossils. • Radioactive decay is at a fixed rate; by comparing the ratio of radioactive and stable isotope, age can be estimated. in a fossil with the • Ratio of Carbon-14 to Carbon-12 is used to date fossils less than 50,000 years old.
Isotopes cont. • 2.Radioactive tracers • Chemicals labelled with radioactive isotopes are used to trace the steps of a biochemical reaction or to determine the location of a particular substance within an organism. • Isotopes of P, N and H were used to determine DNA structure. • Used to diagnose disease. • 3. Treatment of cancer • Can be hazardous to cells.
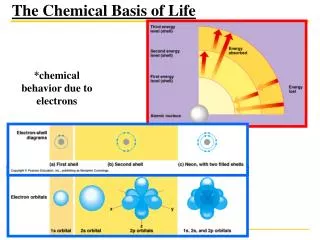
Energy Levels • Electrons are directly involved in chemical reactions. • They have potential energy because of their position relative to the positively charged nucleus. • There is a natural tendency for matter to move to the lowest state of potential energy. • Different fixed potential energy states for electrons are called energy levels or electron shells. • Electrons with lowest potential energy are in energy levels closest to the nucleus. • Electrons with greater energy are in energy levels further from nucleus. • Electrons may move from one energy level to another.
Electron Configuration and Chemical Properties • Electron configuration -- Distribution of electrons in an atom's electron shells; determines its chemical behavior. • Chemical properties of an atom depend upon the number of valence electrons (electrons in the outermost energy level. • Octet rule -- A valence shell is complete when it contains 8 electrons (except H and He). • An atom with an incomplete valence shell is chemically reactive (tends to form chemical bonds until it has 8 electrons to fill the valence shell). • Atoms with the same number of valence electrons show similar chemical behavior.
Bonding in Molecules • Chemical bonds -- Attractions that hold molecules together. • Molecules --Two or more atoms held together by chemical bonds. • Covalent bond -- formed between atoms by sharing a pair of valence electrons; common in organic compounds. • Single covalent bond -- Bond between atoms formed by sharing a single pair of valence electrons. • Double bond -- share two pairs of valence electrons. • Triple bond -- share three pairs of valence electrons. • Compound = A pure substance composed of two or more elements combined in a fixed ratio. • For example: water (H2O), methane (CH4).
Nonpolar Covalent Bonds • Electronegativity -- Atom's ability to attract and hold electrons. • • The more electronegative an atom, the more strongly it attracts shared electrons. • • Scale determined by Linus Pauling: • O = 3.5; N = 3.0; S and C = 2.5; P and H = 2.1. • Nonpolar bond -- Covalent bond formed by an equal sharing of electrons between atoms. • • Occurs when electronegativity of both atoms is about the same. • • Molecules made of one element usually have nonpolar covalent bonds (H2 and O2).
Polar Covalent Bonds • Polar bond -- Covalent bond formed by an unequal sharing of electrons between atoms. • • Occurs when the atoms involved have different electronegativities. • • In water, electrons spend more time around the oxygen than the hydrogens. This causes the oxygen atom to have a slight negative charge and the hydrogens to have a slight positive charge.
Ionic Bonds • Ion -- Charged atom or molecule. • Anion -- An atom that has gained one or more electrons from another atom; negatively charged. • Cation -- An atom that has lost one or more electrons; positively charged. • Ionic bond -- Bond formed by the electrostatic attraction after the complete transfer of an electron from a donor atom to an acceptor. • Strong bonds in crystals, but fragile bonds in water. • Ionic compounds are called salts (e.g. NaCl or table salt).
Biologically important weak bonds • Include: Hydrogen bonds; Ionic bonds in aqueous solutions; Van der Waals forces. • Hydrogen bond -- Bond formed by the charge attraction when a hydrogen atom covalently bonded to one electronegative atom is attracted to another electronegative atom. • Van der Waals -- charge attraction between oppositely charged portions of polar molecules.