Analysis of Various Metal Oxides and Organic Compounds in Environmental Chemistry
This document explores the chemical interactions and transformations of various metal oxides, organic compounds, and anions in environmental contexts. It specifically examines the roles of Fe(III)-citrate, MnO2, and Mn(III)-pyrophosphate, alongside other reactants such as NO3-, U(VI), and DMSO. Multiple supplementary figures illustrate the experimental setups and results for in-depth analysis. The document aims to provide insights into the behavior of these compounds under varying conditions, informing future research in environmental science.


Analysis of Various Metal Oxides and Organic Compounds in Environmental Chemistry
E N D
Presentation Transcript
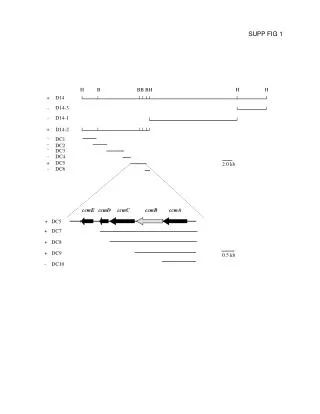
+ DC7 + DC8 + DC9 - DC10 H B BB BH H H + D14 - D14-3 D14-1 - + D14-2 - DC1 - DC2 - DC3 - DC4 + DC5 2.0 kb - DC6 ccmE ccmD ccmC ccmB ccmA DC5 0.5 kb SUPP FIG 1 +
SUPP FIG 2 A. Fe(III)-citrate B. MnO2 C. Mn(III)-pyrophosphate
SUPP FIG 2, Con’d D. NO3- E.g-FeOOH F. U(VI)
SUPP FIG 2, Con’d G. O2 H. DMSO I. TMAO J. Fumarate K. S2O32- L. SO32-
SUPP FIG 3 A. Fe(III)-citrate B. MnO2 C. Mn(III)-pyrophosphate
SUPP FIG 3, Cont’d D. NO3- E.g-FeOOH F. U(VI)
SUPP FIG 3, Cont’d G. O2 H. DMSO I. TMAO J. Fuarate K. S2O32- L. SO32-