Role of Mitochondria in Oxidative Phosphorylation and Cellular Metabolism
This text delves into the process of oxidative phosphorylation, detailing how energy from electrons (NADH and FADH2) is converted into ATP through various complexes (I-IV). It highlights the significance of electron carriers like NAD+, FAD, ubiquinone, cytochromes, and iron-sulfur proteins. The transport mechanisms for NADH to mitochondria and the implications of mitochondrial mutations leading to encephalomyopathies such as Leber’s hereditary optic neuropathy (LHON) and MERRF are discussed. The document also addresses the relationship between mitochondria, apoptosis, and oxidative stress.

Role of Mitochondria in Oxidative Phosphorylation and Cellular Metabolism
E N D
Presentation Transcript
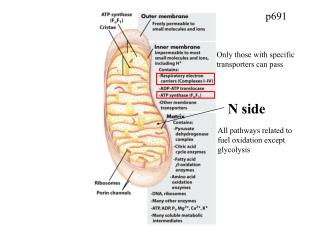
p691 Only those with specific transporters can pass N side All pathways related to fuel oxidation except glycolysis
Oxidative phosphorylation • Converting the energy from electrons (from NADH and FADH2) to ATP • Electron transfer occurs in oxidative phosphorylation: 1. Direct transfer to reduce cation, Fe3+Fe2+ 2. Transfer as hydrogen atom 3. Transfer as hydride ion (:H-)
Five electron carrying molecules 1. NAD+ 2. FAD 3. Ubiquinone 4. Cytochromes 5. Iron-sulfur proteins
p693 Ubiquinone (coenzyme Q; Q) Ubiquinone Plastoquinone (plant chloroplast) Menaquinone (bacteria)
p694 cytochromes
p695 Iron-sulfur proteins Iron-sulfur proteins
p696 Method for determining the sequence of electron carriers
p705 Oxidative phosphorylation
Complex I p698 NADH +H+ FMN Fe2+S CoQ NAD+ FMNH2 Fe3+S CoQH2 NADH +H++Q --> NAD++QH2 4 protons pump out
Complex I & II Succinate FAD Fe2+S CoQ Fumarate FADH2 Fe3+S CoQH2 p697
Complex III p700 CoQH2 cyt b ox Fe2+S cyt c1ox cytcred CoQ cytbred Fe3+S cytc1red cytcox QH2+2Cytc1oxidized+2HN+ --> Q+2Cytc1reduced+4Hp+
Complex IV p702 cyt c red cyt a ox cyt a3red O2 cyt c ox cyt a red cyt a3ox 2 H2O 4Cytcreduced+8HN++O2 --> 4Cytcoxidized+ 4Hp++2H2O
p706 QH2 oxidase (resistant to cyanide)
p707 Chemical uncouplers • Chemicals like DNP and FCCP are weak acid with hydrophobic properties that permit them to diffuse readily across mitochondrial membranes. After entering the matrix in the protonated form, they can release a proton, thus disspating the proton gradient.
p406 Ionophores • Valinomycin (an ionophore) allows inorganic ions to pass easily through membranes. This will uncouple electron transfer from oxidative phosphorylation.
p711 Mitochondrial ATP synthase complex
NADH transport • NADH produced by glycolysis must be transported into mitochondria to produce ATP. • However, NADH cannot enter mitochondria directly. Instead it is transported by the form of malate or glycerol 3-phosphate.
Oxidative phosphorylation in brown fat tissue is uncoupled with ATP synthesis p687
Regulation p718
Mitochondrial genome p720
Mitochondrial encephalomyopathies • Mutations in mitochondrial genes cause mitochondrial encephalomyopathies that affecting primarily the brain and skeletal muscle. Because infants inherit their mitochondria from their mothers, so mitochondrial encephalomyopathies are maternal-linked.
Leber’s hereditary optic neuropathy (LHON) • LHON is the result of defective mitochondrial genes that are involved in electron transfer. • Vision loss usually occurs between the ages of 15 and 35.
Myoclonic epilepsy and ragged-red fiber disease (MERRF) • Mutation in the mitochondrial gene that encodes a tRNA specific for lysine (lysyl-tRNA) results in MERRF. • Synthesis of several proteins require this tRNA is interrupted.
p720 MERRF • MERRF patients often have abnormally shaped mitochondria containing paracrystalline structures. • This lysyl-tRNA mutation is also one of the causes of adult-onset (type II) diabetes.
Mitochondrion is probably evolved from endosymbiotic bacteria p35
Bacteria do have respiratory chain enzymes • For example, E. coli has NAD-linked electron transfer from substrate to O2, coupled to the phosphorylation of cytosolic ADP.
Mitochondria, apoptosis, and oxidative stress Mitochondria is not only involved in ATP synthesis. It is also involved in cellular damage and death.
The role of mitochondria in apoptosis • When cell receives a signal for apoptosis, one consequence is the permeability of the outer mitochondrial membrane will increase, allowing cytochrome c release. • The release of cytochrome c will activate caspase 9, which will initiate the protein degradation process.
Mitochondria and oxidative stress • Antimycin A inhibits complex III by occupying the QN site, which may increase the likelihood of superoxide radical formation and cellular damage.