Alkynes – Chapter 7
Alkynes – Chapter 7 . nomenclature - (chapter 5) , structure, classification acidity of terminal acetylenes - (chapter 4) alkylation prep - dehydrohalogenation (-2HX) or double -elimination reduction - H 2 /M (chapter 6) and chemical hydroboration (chapter 6) - protonation

Alkynes – Chapter 7
E N D
Presentation Transcript
Alkynes – Chapter 7 nomenclature - (chapter 5), structure, classification acidity of terminal acetylenes - (chapter 4) alkylation prep - dehydrohalogenation (-2HX) or double -elimination reduction - H2/M(chapter 6) and chemical hydroboration (chapter 6) - protonation - oxidation (hydration) enol-keto tautomerism addition of HX, X2, H2O (chapter 6) synthesis(chapter 6) (blue- chemistry from previous chapters)
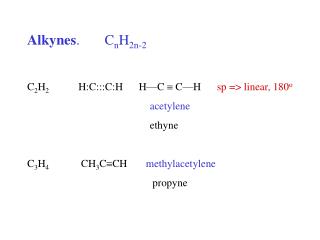
D imethylacetylene V inylacetylene Nomenclature • IUPAC: use the infix -yn- to show the presence of a carbon-carbon triple bond • Common names: prefix the substituents on the triple bond to the word “acetylene” IUPAC name: 2-Butyne 1-Buten-3-yne Common name:
Cycloalkynes • Cyclononyne is the smallest cycloalkyne isolated • it is quite unstable and polymerizes at room temp • the C-C-C bond angle about the triple bond is approximately 155°, indicating high angle strain
Physical Properties • Similar to alkanes and alkenes of comparable molecular weight and carbon skeleton
90o angle bond to orbitals) C R C R’ Acetylene – linear geometry – sp hybridization - 180o bond angles R-C-C-R’ -bond 180o bond angle
Acidity • The pKa of acetylene and terminal alkynes is approximately 25, which makes them stronger acids than ammonia but weaker acids than alcohols (Section 4.1) • terminal alkynes react with sodium amide to form alkyne anions
H H H H H C C C H > CH C > C C 3 CH 3 CH H 3 H H pKa = 50 pKa = 44 H-Csp3 H-Csp2 The more s-character,the moreacidic the C-H bond 50% 33% 25% Acidity of organic compounds pKa = 25 H-Csp Why ? recall chapter 4
pKa = 16 also -NH2 H-NH3 pKa = 38 Relative basicity (acidity)
Acidity • terminal alkynes can also be converted to alkyne anions by reaction with sodium hydride or lithium diisopropylamide (LDA) • because water is a stronger acid than terminal alkynes, hydroxide ion is not a strong enough base to convert a terminal alkyne to an alkyne anion
Alkylation of Alkyne Anions • Alkyne anions are both strong bases and good nucleophiles • They participate in nucleophilic substitution reactions with alkyl halides to form new C-C bonds to alkyl groups; they undergo alkylation • because alkyne anions are also strong bases, alkylation is practical only with methyl and 1° halides • with 2° and 3° halides, elimination is the major reaction
Alkylation of Alkyne Anions • alkylation of alkyne anions is the most convenient method for the synthesis of terminal alkynes • alkylation can be repeated and a terminal alkyne can be converted to an internal alkyne
Preparation from Alkenes • Treatment of a vicinal dibromoalkane with two moles of base, most commonly sodium amide, results in two successive dehydrohalogenation reactions (removal of H and X from adjacent carbons) and formation of an alkyne Note! alkenes are precursors of diBrs dehydrohalogenation
Preparation from Alkenes • for a terminal alkene to a terminal alkyne, 3 moles of base are required
N a N H 2 R C – C = C R - H B r R C C C R A haloalkene (a vinylic halide) Preparation from Alkenes • a side product may be an allene, a compound containing adjacent carbon-carbon double bonds, C=C=C R H C C C X H H R R An allene H R R An alkyne
Allene • Allene: a compound containing a C=C=C group • the simplest allene is 1,2-propadiene, commonly named allene
Allenes • most allenes are less stable than their isomeric alkynes, and are generally only minor products in alkyne-forming dehydrohalogenation reactions
R' Br R' Br R' Br C Br C 2 C Br 2 1 eq. C 1 eq. C C Br or XS Br R R R Br Electrophilic Addition of X2 • Alkynes add first mole of bromine to give a dibromoalkene (2nd mole give tetrabromoalkane) • addition shows anti stereoselectivity
Addition of X2 • the intermediate in bromination of an alkyne is a bridged bromonium ion
B r B r H B r H B r C H C C H C H C = C H C H C C H 3 3 2 3 3 B r Addition of HX • Alkynes undergo regioselective addition of either 1 or 2 moles of HX, depending on the ratios in which the alkyne and halogen acid are mixed Propyne 2-Bromopropene 2,2-Dibromopropane
Addition of HX • the intermediate in addition of HX is a 2° vinylic carbocation • reaction of the vinylic cation (an electrophile) with halide ion (a nucleophile) gives the product
Addition of HX • in the addition of the second mole of HX, Step 1 is reaction of the electron pair of the remaining pi bond with HBr to form a carbocation • of the two possible carbocations, the favored one is the resonance-stabilized 2° carbocation
Hydroboration Acetylene chemistry - recall olefin chemistry R = H, alkyl
Hydroboration • Addition of borane to an internal alkyne gives a trialkenylborane • addition is syn stereoselective
Hydroborationof an internal alkyne of a terminal alkyne – get dihydroboration
Hydroboration • to prevent dihydroboration with terminal alkynes, it is necessary to use a sterically hindered dialkylborane, such as (sia)2BH • treatment of a terminal alkyne with (sia)2BH results in stereoselective and regioselective hydroboration
Hydroboration Terminal acetylene hindered borane adds once di-sec-isoamylborane or HB(sia)2 tautomerism H+ transfer (O to C)
enol-keto tautomerismH+ transfer C O [from hydroboration, oxymercuration (next) and other rxns] Enol Ketone mechanism e(-) arrows or
Hydroboration • Treating an alkenylborane with H2O2 in aqueous NaOH gives an enol • enol:a compound containing an OH group on one carbon of a carbon-carbon double bond • an enol is in equilibrium with a keto form by migration of a hydrogen from oxygen to carbon and the double bond from C=C to C=O • keto forms generally predominate at equilibrium • keto and enol forms aretautomersand their interconversion is called tautomerism
1 . B H 3 2 . H O , N a O H 2 2 Hydroboration • hydroboration/oxidation of an internal alkyne gives a ketone • hydroboration/oxidation of a terminal alkyne gives an aldehyde O 3-Hexyne 3-Hexanone
O H H S O 2 4 C H C C H C H C C H H O C H C = C H 3 3 3 2 3 2 H g S O 4 1-Propen-2-ol Propanone (an enol) (Acetone) Addition of H2O: hydration • In the presence of sulfuric acid and Hg(II) salts, alkynes undergo addition of water O + Propyne
Addition of H2O: hydration • Step 1: attack of Hg2+ (an electrophile) on the triple bond (a nucleophile) gives a bridged mercurinium ion • Step 2: attack of water (a nucleophile) on the bridged mercurinium ion intermediate (an electrophile) opens the three-membered ring
Addition of H2O: hydration • Step 3: proton transfer to solvent gives an organomercury enol • Step 4: tautomerism of the enol gives the keto form
Addition of H2O: hydration • Step 5: proton transfer to the carbonyl oxygen gives an oxonium ion • Steps 6 and 7: loss of Hg2+ gives an enol; tautomerism of the enol gives the ketone
Hydration of acetylenes terminal yne - HBR2/[O] aldehyde - Hg++/H2O ketone internal yne - “either” ketone(s)
Reduction • Treatment of an alkyne with hydrogen in the presence of a transition metal catalyst, most commonly Pd, Pt, or Ni, converts the alkyne to an alkane
Reduction • With the Lindlar catalyst, reduction stops at addition of one mole of H2 • this reduction shows syn stereoselectivity
Hydroboration - Protonolysis • Addition of borane to an internal alkyne gives a trialkenylborane • addition is syn stereoselective • treatment of a trialkenylborane with acetic acid results in stereoselective replacement of B by H
Dissolving Metal Reduction • Reduction of an alkyne with Na or Li in liquid ammonia converts an alkyne to an alkene with anti stereoselectivity
Dissolving Metal Reduction • Step 1: a one-electron reduction of the alkyne gives a radical anion • Step 2: the alkenyl radical anion (a very strong base) abstracts a proton from ammonia (a very weak acid)
+ N a N a Dissolving Metal Reduction • Step 3: a second one-electron reduction gives an alkenyl anion • this step establishes the configuration of the alkene • a trans alkenyl anion is more stable than its cis isomer • Step 4: a second acid-base reaction gives the trans alkene R R . : + + C C C C R R H H An alkenyl anion
Organic Synthesis : Beginning Concepts • A successful synthesis must • provide the desired product in maximum yield • have the maximum control of stereochemistry and regiochemistry • do minimum damage to the environment (it must be a “green” synthesis) • Our strategy will be to work backwards from the target molecule
Organic Synthesis : Beginning Concepts • We analyze a target molecule in the following ways • the carbon skeleton: how can we put it together. Our only method to date for forming new a C-C bond is the alkylation of alkyne anions (Section 7.5) • the functional groups: what are they, how can they be used in forming the carbon-skeleton of the target molecule, and how can they be changed to give the functional groups of the target molecule
target starting molecule materials Organic Synthesis : Beginning Concepts • We use a method called a retrosynthesis and use an open arrow to symbolize a step in a retrosynthesis • Retrosynthesis: a process of reasoning backwards from a target molecule to a set of suitable starting materials
Organic Synthesis : Beginning Concepts H CH Br H 2 C C H H C H 3 How does one make butanal from 1-bromobutane? O H H C H C C H C H H 3
H H CH Br 2 C C H H C H 3 H H CH OH 2 C C H H C H 3 O H H C H C C H C H H 3 Organic Synthesis : Beginning Concepts NaOH DMF PCC
Organic Synthesis : Beginning Concepts • Target molecule: cis-3-hexene
Organic Synthesis : Beginning Concepts • starting materials are acetylene and bromoethane
Organic Synthesis : Beginning Concepts • Target molecule: 2-heptanone