Ionic and Covalent Bonds | Types, Formation, and Characteristics Explained
Explore the fundamental concepts of ionic and covalent bonding, including the process of bond formation, characteristics of each bond type, and examples of compounds. Learn about properties such as melting points, conductibility, and multiple bond structures.

Ionic and Covalent Bonds | Types, Formation, and Characteristics Explained
E N D
Presentation Transcript
Bonds Between Atoms Ionic Covalent Network Solids Molecular Substance
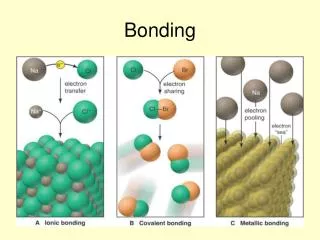
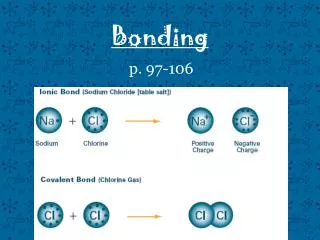
Ionic Bonds • What is an Ionic Bond? • - An Ionic Bond is a chemical bond resulting from the TRANSFER of electrons from one bonding atom to another • When is an ionic bond formed? - An ionic bond is formed when a cation (positive ion) transfers electrons to an anion (negative ion).
Example 1 www.mhhe.com/physsci/chemistry/animations/chang_7e_esp/bom1s2_11.swf -
Example 2 • Some atoms, such as oxygen, need to gain two electrons to achieve stability. • The two electrons released by one magnesium atom could be gained by a single atom of oxygen. • When this happens, magnesium oxide (MgO) is formed.
Potassium Oxide • Click for Animation
Covalent Bonds • What is an Covalent Bond? • - A covalent bond is a chemical bond resulting from SHARING of electrons between 2 bonding atoms. • What forms a covalent bond? - A covalent bond is formed between two nonmetals. Click for animation
Examples of Covalent molecules • The neutral particle is formed when atoms share electrons is called a molecule • A molecule is the basic unit of a molecular compound.
What are some characteristics of an ionic bond? • Crystalline at room temperatures • Have higher melting points and boiling points compared to covalent compounds • Conduct electrical current in molten or solution state but not in the solid state
What are some characteristics of a covalent bond? • Covalent bonds have definite and predicable shapes. • Very strong • Low melting and boiling points
Covalent Bonds can have multiple bonds, so you should be familiar with the following… Single Covalent Bond- chemical bond resulting from sharing of an electron pair between two atoms. Double Covalent Bond- chemical bond resulting from sharing of two electron pairs between two atoms. Triple Covalent Bond-chemical bond resulting from sharing of three electron pairs between two atoms.
Covalent Network Structures • A covalent network is when atoms are bonded together covalently to form giant macromolecular structures. • The element Carbon can form Covalent Network structures • Two examples are: Diamond & Graphite
Diamond • Each Carbon atom bonds to 4 others • Strong covalent bonds throughout • High melting points and Boiling points • No free electrons so does not conduct electricity.
Graphite • Each Carbon atom is bonded to 3 others • The bonding within the layers is strong covalent. • The spare fourth electron of each C atom is delocalised so graphite can conduct electricity • There are weak intermolecular forces of attraction between the layers.
Covalent Networks • Another example of a covalent network is silicon dioxide. This is a compound though it resembles diamond.