The Mole
The Mole. What is it? How do we use it?. Calculating the mass of atoms:. Carbon-12 was used as the standard for relative mass. A single atom of C-12 is assigned a mass of exactly 12 atomic mass units . The symbol for atomic mass units is u or amu .

The Mole
E N D
Presentation Transcript
The Mole What is it? How do we use it?
Calculating the mass of atoms: • Carbon-12 was used as the standard for relative mass. • A single atom of C-12 is assigned a mass of exactly 12 atomic mass units. • The symbol for atomic mass units is u or amu. • The mass of an atom expressed in atomic mass units is called the atomic mass of the atom.
Calculation of Formula Weight • Formula weight- sum of the atomic weight of each atom in the chemical formula. • Ex. Formula wt. of NaOH • FW = 1 (atomic weight of Na) + 1 (atomic weight of O) + 1 (atomic weight of H)= 22.989770 + 15.9994 + 1.00794 = 39.9971 amu
More Practice • Ex. Formula wt. of Al(OH)3 • 1 amu Al + 3 amu O + 3 amu H = 26.981538+3(15.9994)+3(1.00794) =78.0036 amu
Measuring Amounts • We often measure the amounts of things by one of 3 methods: • By count • By mass • By volume • For example, you can by soda by the six-pack or the liter.
We can buy oranges by count or by mass (weight). Ex. Oranges: 8 for $3.00 or $.79/lb It’s easy to buy big items by count: apples, oranges, pens, etc. but sometimes it’s easier to buy them when they are grouped together: Ex. A dozen eggs, a gross of pencils… what else? What about smaller items? How about grains of sand in a timer?
The Mole • Chemists use a number that is a specified number of particles to represent particles that are too small to count individually. It’s the Mole
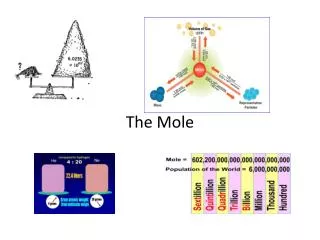
The Mole • 1 mole is: 602,200,000,000,000,000,000,000 or 6.022 x 1023 representative particles A representative particle means ITEMS: atoms, molecules, formula units
Molar Mass (aka Molecular Weight) • Molar mass (also known as molecular weight) - mass of one mole something. • Scientists made it easy by making molar mass in grams the same number as atomic mass in amu. • Units of molar mass: grams per mole. (g/mol)
What is the mass of one mole of aluminum? 26.981538 g
Practice • What is the mass of 2.3 moles of sulfur? • Known: S mm is 32.066 g/mol. • Unknown: mass (in grams) • Start with what we know: • 2.3 mol S 32.066 g 1 mol
2.3 mol S 32.066 g S 1 mol Set up the problem so that the unit you want is on the top • Cross out the units that cancel and then multiply 2.3 mol S = 73.752 g
What if I know the grams and want to know the # of moles? • How many moles are in 412 grams of CaSO4? • Known: have 412 g. CaSO4 Molar mass of CaSO4 = 136.142 g/mol 412 g 1 mol CaSO4 136.146 g =3.03 mol CaSO4
Practice • How many grams are in 2.4 moles of NaCl?
A little background on Chemical Reactions… • Reactant 1 + Reactant 2 Product 1 + Product 2 • Reactants and products can be elements and/or compounds • (it depends on the type of reaction….)
Synthesis Reactions • Two or more substances combine to form a single substance • Reactants: 2 substances • Product: Always 1 compound
A General Formula A + B AB
Synthesize Your Own! • Describe a “real world” example of a synthesis reaction + Marriage
Decomposition Reactions • Opposite of synthesis reaction • A single compound is broken down into 2 or more products • Require energy • Heat, light, electricity
Decomposition Reactions • What are the reactants and products? • Reactant: 1 Compound • Products: 2 or more Elements / Compounds
A General Formula AB A + B
Decompose Your Thoughts! • Describe a “real world” example of a decomposition reaction + Break-up
Combustion Reactions • Always a substance reacting with O2to release energy • Most common is the reaction of a hydrocarbon with O2
A General Formula CxHy + O2 CO2 + H2O
COMBUSTIONrxnexamples • C3H8 + O2 CO2 + H2O • C4H10 + O2 CO2 + H2O • C6H6 + O2 CO2 + H2O • NH3 + O2 NO + H2O
Single Replacement Reactions • A single uncombined element replaces an element in a compound • Reactants: • 1 Element and 1 compound • Products: • 1 Different element and 1 different compound
A General Formula A + BC AC + B
Replace with Simplicity • Describe a “real world” example of a single displacement reaction + + “Cutting In” on a Dance
Double Replacement Reactions • The cations and anions of two different compoundsswitch places • Reactant: Two ionic compounds • Product: Two different ionic compounds
A General Formula AB + CD AD + CB • Cations: A and C • Anions: B and D
Replace with Simplicity • Describe a “real world” example of a double replacement reaction Switching dance partners
How to Determine Products • Identify cations and anions in each compound • Pair each cation with the anion from the other compound • Write 2 new formulas for the products • Use ion sheets!
Types of Products in Double Replacement Reactions • Precipitate (solid) • Not soluble in the solution • Gas • Molecular compound • Made of 2 nonmetals • Ex. Water
Homework • Summary of Reaction Types WS
Synthesis Predict the Products • Mg + F2 • Ba + O2 • Li + Br2
DecompositionPredict the Products • H2O • MgCl2 • FeS
Decompose Your Thoughts! • Describe a “real world” example of a decomposition reaction • Example: Divorce
Opposites Remember? • Create a T-chart • On the left side, write in the general equation, reactants, and rules of predicting the products of synthesis reactions • On the right side, write in the general equation, reactants, and the rules of predicting the products of decomposition reactions