The Phosphorus Cycle
The Phosphorus Cycle. Masta B Yoo, Devin Kim, Drew Li, Justin Yun, Raymond Kim . Basic Chemistry. Essential element of life Phosphorus is a Group 15 (5A) element Symbol: P Atomic number: 15 Atomic mass: 30.974 Color: colorless/ red/ silvery white. Basic Chemistry continued. Nonmetal

The Phosphorus Cycle
E N D
Presentation Transcript
The Phosphorus Cycle Masta B Yoo, Devin Kim, Drew Li, Justin Yun, Raymond Kim
Basic Chemistry • Essential element of life • Phosphorus is a Group 15 (5A) element • Symbol: P • Atomic number: 15 • Atomic mass: 30.974 • Color: colorless/ red/ silvery white
Basic Chemistry continued • Nonmetal • Density: 1823 kg/m3 • Atomic radius: 100 pm • Solid • Melting Point: 317.3 K • Boiling Point: 550 K
Basic Chemistry continued • Key ingredient to plants fertilizers • In nature, Phosphorus exists in form of Phosphate
Basic Chemistry, cont. • Formulas of rocks containing phosphate are fluoroapatite (3Ca3(PO4)2.CaF2), chloroapaptite, (3Ca3(PO4)2.CaCl2), and hydroxyapatite (3Ca3(PO4)2.Ca(OH)2)
Description of Phosphorus • Most common form of phosphorus is red and white. • Both are tetrahedral groups of four atoms. • White phosphorus burns on contact with air and can change into red phosphorus when exposed to light or heat.
Description of Phosphorus Cont. • White phosphorus has two modifications. • Ordinary phosphorus is waxy white solid. • When pure it is colorless and transparent.
Description, Cont. • Pure phosphorus is insoluble in water but soluble in carbon disulphide • Catches fire spontaneously in air.
Background • Very poisonous • Never found in free nature • Essential ingredient of all cell protoplasm, nervous tissue, and bones • Most commonly found as phosphates • Part of DNA materials and energy distribution
Uses of Phosphorus • Used to make waterproof matches, pyrotechnics, smoke screens, tracer bullets, and incendiary shells. • Used in fertilizer • Used to produce special glasses, for example sodium lamps
Uses, continued • Calcium Phosphate is used to create fine Chinaware • Monocalcium Phosphate used to make baking powder • Used in making steel, and Phosphor Bronze • Na3PO4 is used as a cleaning agent, water softener, and also prevents corrosion in pipes
Hazards and Risks • Severely toxic • White form is more toxic than red form • Chronic poisoning leads to necrosis of jaw (phossy-jaw) • Phosphate pollution comes from detergents, insecticides, and fertilizers • Inorganic phosphorus is harmless
Background • The phosphorus cycle does not have a gas phase, but small amounts of phosphoric acid rise into the atmosphere, contributing to acidic precipitation.
Background • The water, carbon, and sulfur cycles all include at least one phase in which the element is in its gaseous state. The largest reservoir of phosphorus is in sedimentary rock.
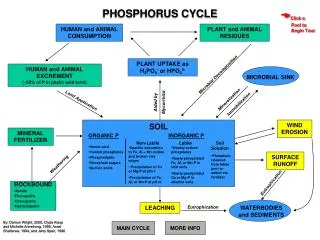
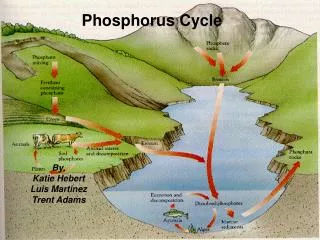
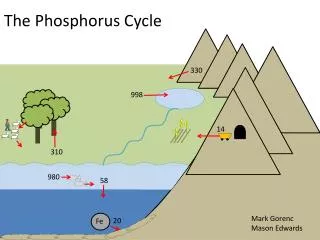
Description of Cycle • Simplest of the cycles • Phosphorus has only one form – phosphate • Always part of an organism, dissolved in water, or in the form of rock
Description of Cycle • Phosphorus enters environment from rocks or deposits • Apatite is the phosphate rock where phosphate is available • Weathering and erosion releases phosphate ions that are soluble in water
Description, cont. • Phosphate then acts as fertilizers or nutrients for land plants • It gets incorporated into molecules essential for life like ATP, adenosine triphosphate – this is important in storage and use of energy
Description, cont. • Phosphate is in backbone of DNA and RNA • When animals and plants die, phosphates will return to the soils or oceans again during decay
Description, cont. • Phosphorus cycles through plants and animals much faster than it does through rocks and sediments.. • A lot of phosphate goes into the water from erosion and leaching • Water plants use this phosphate as nutrients
Description, cont. • Phosphate is the limiting agent in growth of plants and algae • If there is a lack of phosphate, plants grow slowly or are stunted • If too much, excess growth may occur
Description, cont. • Phosphate in water is precipitated from water as ion phosphate (insoluble) • Phosphate in shallow sediment may be recycled into water • In deep sediment in water, it is available as part of as part of rock formations for cycle to repeat
Description, cont. • After that, phosphorus will end up in sediments or rock formations again, remaining there for millions of years. • Eventually, phosphorus is released again through weathering and the cycle starts over.
Human Influences on Cycle • Human influences on phosphate cycle mostly comes from introduction & use of commercial synthetic fertilizers. • Phosphate obtained by mining at certain deposits of calcium phosphate called apatite.
Influences cont. • Phosphate rock and sulfuric acid are combined to make a fertilizer called “super phosphate.” • There are many negative effects of using this fertilizer.
Influences cont. • A lot of the fertilizer is lost through the water run-off because plants aren’t able to use up all of it. • Eventually the wasted phosphate in the water is precipitated as it settles at the bottom of the body of water.
Influences cont. • In some lakes & ponds the phosphate can be recycled as a problem nutrient. • Other sources of phosphate are in out flows from local sewage treatment plans.
Interesting Facts • Originally, phosphorus was extracted from urine. • Highly energy intensive. • Gr. phosphoros, meaning “light bearer”
Facts, cont. • In 1669, German alchemist Henning Brand discovered phosphorus from a preparation from urine. • The only correct spelling of the element is phosphorus. There does exist a word phosphorous, but it is the adjectival form for the smaller valency
Bibliography • “Nutrient Cycling in Ecosystems.” http://home.earthlink.net/~dayvdanls/ecosys4.htm 17 September 2004 • “Phosphorus.” http://pearl1.lanl.gov/periodic/elements/15.html 17 September 2004
Bibliography cont. • “Web-Elements.” http://www.webelements.com/webelements/elements/text/P/key.html 17 September 2004 • http://en.wikipedia.org/wiki/Phosphorus 17 September 2004 • “Virtual Chembook.” http://www.elmhurst.edu/~chm/vchembook/308phosphorus.html 17 September 2004