Understanding the Electromagnetic Spectrum: Energy, Frequency, and Wavelength Calculations
120 likes | 225 Vues
This lesson covers the electromagnetic spectrum, focusing on the relationships between energy, frequency, and wavelength of light. Students will learn to calculate these properties using Planck’s constant and the speed of light. The lesson emphasizes the characteristics of electromagnetic radiation, including visible light, and discusses wave properties such as wavelength and frequency. Students will practice calculations that illustrate the inverse relationship between these quantities, as well as the direct relationship between energy and frequency.

Understanding the Electromagnetic Spectrum: Energy, Frequency, and Wavelength Calculations
E N D
Presentation Transcript
Lesson Objectives • Explore the electromagnetic spectrum (6B) • Understand the mathematical relationships between energy, frequency, and wavelength of light (6B) • Calculate the wavelength, frequency, and energy of light using Planck’s constant and the speed of light (6C) • Electromagnetic Spectrum and Quantized Energy Calculations
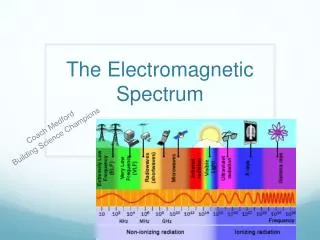
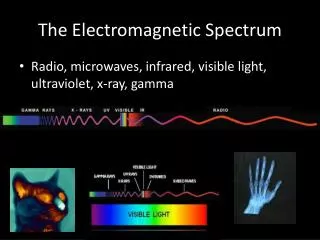
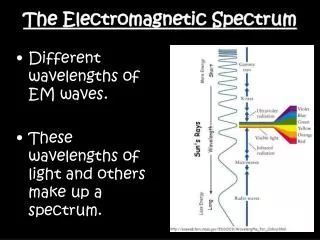
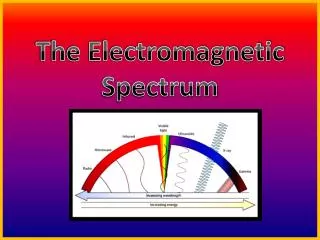
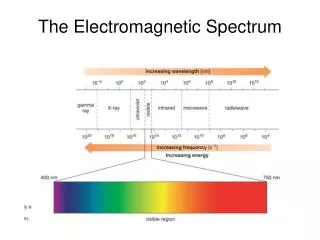
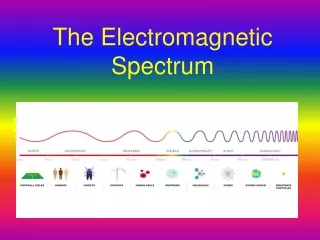
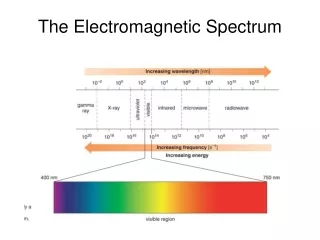
Electromagnetic spectrum includes a wide range of radiation • Electromagnetic radiation – form of energy that displays characteristics of a wave as it moves through space • Electromagnetic Spectrum of Radiation
Visible light is type of electromagnetic radiation • Comprises a small portion of the electromagnetic spectrum • Ranges from 400–700 nm • Visible Light User:Tatoute [GFDL ]
Waves are characterized by • Wavelength(λ) – distance between two successive peaks • Units of meters (m) • Frequency ( f ) – number of wave cycles completed during a unit of time • Units of hertz (Hz) Wave diagram • Wave Nature of Light
Wavelength and frequency are inversely proportional • Mathematical association between wavelength and frequency expressed in relation to speed of light (c) c = 3.00 x 108 m/s • Wave Nature of Light Wave diagram
Wave Calculation • Write unknown and givens λ= 5.50 x 10-6 m c = 3.00 x 108 m/s f = ? • Identify the formula and rearrange, if needed • Convert units and find intermediates, if needed Ex) What is the frequency of a photon whose wavelength is 5.50 x 10-6 m? • Plug in and solve • Make sure the answer is reasonable
Energy of waves is • Directly proportional to frequency • Inversely proportional to wavelength • Energy of Waves Energy
Wave model of light did not fully explain the behavior of light • Only particular frequencies of light are absorbed or emitted by matter in certain situations • Short Comings of the Wave Model Absorption Spectrum Emission Spectrum
Energy is absorbed and released by particles in small discrete amounts • Quantum – small, fixed amount of energy • Mathematical equation that describes the relationship between energy and frequency Ephoton= hf • Energy (E) is in units of joules • Planck's constant (h) is equal to 6.63 x 10-34 J∙s • Frequency ( f ) is in units of s-1 or Hz • Energy is directly proportional to frequency • Quantum Theory
Energy & Frequency Calculation • Write unknown and givens f = 6.26 × 1014 Hz Ephoton= ? h = 6.63 × 10–34 J∙s • Identify the formula and rearrange, if needed • Convert units and find intermediates, if needed Ex) Light with a frequency of 6.26 × 1014 Hz has photons that carry how much energy? • Plug in and solve • Make sure the answer is reasonable
Wavelength and frequency are inversely proportional c = λf where c = 3.00 x108 m/s • Frequency is directly proportional to energy Ephoton= hf where h = 6.63 x 10-34 J∙s • Combining the formulas creates an equation that shows the inverse relationship between wavelength and energy • Rearrange c =λf to solve for f • Substitute c/λfor f in E = hf • Energy, Frequency, and Wavelength
Energy & Wavelength Calculation • Write unknown and givens Ephoton= ? λ=2.21 × 10-7 m c = 3.00 × 108 m/s h = 6.63 × 10–34 J∙s • Identify the formula and rearrange, if needed • Convert units and find intermediates, if needed Ex) What is the energy of a photon whose wavelength is 2.21 x 10-7 m? • Plug in and solve • Make sure the answer is reasonable