100
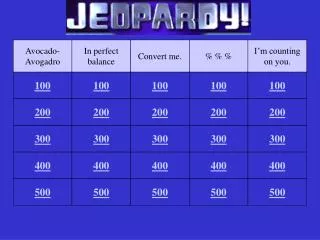
Avocado- Avogadro. In perfect balance. Convert me. % % %. I’m counting on you. 100. 100. 100. 100. 100. 200. 200. 200. 200. 200. 300. 300. 300. 300. 300. 400. 400. 400. 400. 400. 500. 500. 500. 500. 500. What is Avogadro’s number?. Column 1, 100.

100
E N D
Presentation Transcript
Avocado- Avogadro In perfect balance Convert me. % % % I’m counting on you. 100 100 100 100 100 200 200 200 200 200 300 300 300 300 300 400 400 400 400 400 500 500 500 500 500
What is Avogadro’s number? Column 1, 100
How many moles are in 3.31 x 1024 molecules of strontium carbonate (SrCO3)? Column 1, 200
How many atoms are in 17.0 moles of silver (Ag)? Column 1, 300
How many moles of magnesium are 1.25 x 1023 atoms of magnesium? Column 1, 400
How many atoms are in 3.65 moles of glucose (C6H12O6)? Column1, 500
State the law of conservation of matter. Column 2, 100
Complete an atom inventory for the following equation, then determine if the equation is balanced or not. Li2O + H2O 2 LiOH Column 2, 200
In order to balance this equation, what coefficient must be placed in front of water? Fe2O3 + H2 → Fe + H2O Column 2, 300
Show the coefficients used to balance the following chemical equation: H3PO4 + HCl → PCl5 + H2O Column 2, 400
Show the coefficients used to balance the following chemical equation: SeCl6 + O2→ SeO2 + Cl2 Column 2, 500
How many moles are in 33 grams of gold (Au)? Column 3, 100
How many grams are in 7.0 moles of CaSO4? Column 3, 200
How many moles are in 594.8 grams of (NH4)2SO4? Column 3, 300
How many moles are present in 352 g of iron (III) oxide, Fe2O3? Column 3, 400
How many grams are present in 1.43 moles of magnesium hydroxide, Mg(OH)2? Column 3, 500
What percent of glucose (C6H12O6) is oxygen? Column 4, 100
What percent of CuCl2 is chlorine? Column 4, 200
What percent of Al(NO3)3 is nitrogen? Column 4, 300
What percent of iron(III) hydroxide, Fe(OH)3, is oxygen? Column 4, 400
Which element makes up the greatest percentage of Al(NO3)3? What percent is that? Column 4, 500
Calculate the molar mass of C2H4 Column 5, 100
How many atoms are in 3 molecules of AgNO3? Column 5, 200
Calculate the molar mass of Zn3(PO4)2 Column 5, 300
Find the molar mass of malachite: Cu2CO3(OH)2. Column 5, 400
What is the molar mass of Paris Green? Cu(C2H3O2)2·3Cu(AsO2)2 Column 5, 500