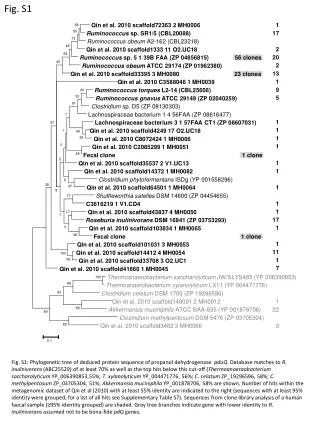
1.Lect01_Ch1
Physics I- Ch1

1.Lect01_Ch1
E N D
Presentation Transcript
Chapter 1 Units, Physical Quantities and Vectors Lecture 1 8/19/2017 CTP- PHYS-1110
Lecture Outline 1.1 The Nature of Physics 1.3 Standards and Units 1.4 Unit Consistency and Conversions 1.5 Uncertainty and Significant Figures 8/19/2017 PHYS-1110
By the end of this lecture students will be able to: o Define the standards for SI units o Convert units of physical quantities to SI units o Use of significant figures in calculations 8/19/2017 PHYS-1110
Introduction Q. Why Study Physics? ? Physics is crucial to understanding the world around us, the world inside us, and the world beyond us. It is the most basic and fundamental science. ? Physics is everywhere, appearing throughout our “day-to-day” experiences. ? If You Want to Know How and Why? Learn Physics 8/19/2017 PHYS-1110
1.1 The nature of physics • Physics is an experimental science in which physicists study the laws of nature. Its principles underlie all the basic sciences, such as astronomy, biology, chemistry and geology., the purpose is to find physical laws that relate the phenomena of nature. • A very well established or widely used theory is called a physical law or principle. 8/19/2017 PHYS-1110
1.2 Standards and Units ? Measurements is the Basis of testing theories in science ? Any measured quantity in nature is a physical quantity ? A physical quantity is specified by a number (the size of the quantity) and a unit and sometimes direction ? In nature, we have fundamental (basic) physical quantities like length, mass and time (can not be derived form other physical quantities) ? All other physical quantities can be derived from the fundamental quantities Ex: speed can be derived from length and time 8/19/2017 PHYS-1110
Units ? Need to have consistent systems of units for the measurements Standardized systems ? agreed upon by some authority, usually a governmental body ? SI -- Systéme International ? agreed to in 1960 by an international committee ? main system used in this course 8/19/2017 PHYS-1110
?Length ? Units: SI – meter, m ? Defined in terms of a meter – the distance traveled by light in a vacuum during a given time. ?Mass ? Units: SI – kilogram, kg ? Defined in terms of a kilogram, based on a specific cylinder kept at the International Bureau of Standards. ?Time ? Units: seconds, s ? Defined in terms of the oscillation of radiation from a cesium atom. 8/19/2017 PHYS-1110
8/19/2017 PHYS-1110
Prefixes ? Prefixes powers of 10 ? Each prefix has a specific name and abbreviation ? The prefixes can be used with any base units ? They are multipliers of the base unit ? Examples: ? 1 mm = 10-3m ? 1 mg = 10-3g correspond to 8/19/2017 PHYS-1110
Prefixes ?Unit prefixes size the unit to fit the situation. 8/19/2017 PHYS-1110
1.4 Unit consistency and conversions • An equation must be dimensionally consistent. Terms to be added or equated must always have the same units. • Always carry units through calculations. • Convert to standard units as necessary. ? When you Multiply or Divide measurements you must carry out the same operation with the units as you do with the numbers 50 cm x 150 cm = 7500 cm2 20 m / 5 s = 4 m/s or 4 ms-1 16m / 4m = 4 • When you Add or Subtract measurements they must be in the same units and the units remain the same 50 cm + 150 cm = 200 cm 20 m/s – 15 m/s = 5 m/s 8/19/2017 PHYS-1110
Conversion of Units ? When units are not consistent, you may need to convert to appropriate ones ? Units can be treated like algebraic quantities that can cancel each other out ? Example ? 15 0 . in ? cm . 2 ? ? 54 cm ? ? ? 15 0 . in 38 1 . cm ? ? 1 in 8/19/2017 PHYS-1110
Examples of various units measuring a quantity 8/19/2017 PHYS-1110
8/19/2017 CTP_PHYS-1110
Q1: Convert 50.0 km/hr to standard units (m/s) km km 1000 m hr 1 m ? ? ? ? 50 0 . 50 0 . 13 9 . hr hr km 1 3600 s s Q2. One gallon of paint (volume = 3.78×10–3m3) covers an area of 25.0 m2. What is the thickness of the paint on the wall in micron? A: 151 µm 8/19/2017 PHYS-1110
1.5 Uncertainty and Significant Figures • A measurement always has some degree of uncertainty. • The length of the nail is 2.8 cm • The length of nail is: 2.85 cm More accurate measurement Less uncertainty • The uncertainty of a measured quantity is indicated by its number of significant figures. Definition: Significant figures are the meaningful digits in the measurements 8/19/2017 PHYS-1110
8/19/2017 CTP_PHYS-1110
How Many Significant Figures? 102 1422 102.0 65,321 1.02 1.004 x 105 0.00102 200 0.10200 1.02 x 104 435.662 1.020 x 104 50.041 8/19/2017 PHYS-1110
Rules for counting Significant figures 1. Nonzero integers always count as significant figures. 1457 has 4 significant figures 23.3 has 3 significant figures 2. Zeros a.Leading zeros - never count 0.0025 2 significant figures b.Captive zeros - always count 1.008 4 significant figures c.Trailing zeros - count only if the number is written with a decimal point 100 1 significant figure 100. 3 significant figures 120.0 4 significant figures 8/19/2017 PHYS-1110
Rules for Multiplication and Division 8/19/2017 PHYS-1110
8/19/2017 CTP_PHYS-1110
Multiplication and Division 32.27 ? 1.54 = 49.6958 49.7 3.68 ? .07925 = 46.4353312 46.4 1.750 ? 0.0342000 = 0.05985 0.05985 3.2650?106? 4.858 = 1.586137 ? 107 1.586 ? ?107 6.022?1023? 1.661?10-24= 1.000000 1.000 8/19/2017 PHYS-1110
8/19/2017 PHYS-1110
Calculate the following. Give your answer to the correct number of significant figures and use the correct units 176 km 11.7 km x 15.02 km = 3900 mm 12 mm x 34 mm x 9.445 mm = 2 m/s 14.05 m / 7 s = 108 kg / 550 m3= 0.2 kg/m 37 L 23.2 L + 14 L = 67.1 s 55.3 s + 11.799 s = 12.2 cm 16.37 cm – 4.2 cm = 116.20 km 350.55 km – 234348 m = 8/19/2017 PHYS-1110
Q1. The speed of a car is 85 mile/h. Express this mile 1609 ? 1 m speed in meters per second. 8/19/2017 PHYS-1110
Q2. The volume of the world's largest cut diamond is 1.84 cubic inches. What is its volume in; ? 1 inch . 2 54 cm 1. cubic centimetres? 2. cubic meters? 8/19/2017 PHYS-1110