Basic Organic Chemistry II
Review of Carbon Atom.

Basic Organic Chemistry II
E N D
Presentation Transcript
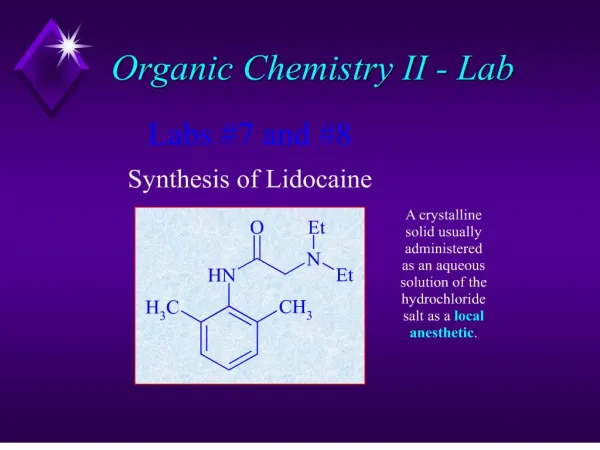
Organic Chemistry Review Part II
Functional Groups Hydrocarbons Derivatives of Hydrocarbons
Functional Groups • Organic molecules may have functional groups attached. • A functional group is a group of atoms of a particular arrangement that gives the entire molecule certain chemical characteristics. • Functional groups are named according to the composition of the group.
Functional Groups • Organic chemists use the letter "R" to indicate an organic molecule. • The "R" can be any organic molecule.
Hydrocarbons • The simplest organic compounds. • Contain only carbon and hydrogen, • Can be straight-chain, branched chain, or cyclic molecules. • Carbon tends to form four bonds in a tetrahedral geometry.
Hydrocarbons • Two classifications: • Aliphatics • Aromatics • Aliphatic - hydrocarbons which do not contain an aromatic ring.
Hydrocarbons • Aromatic- Aromatic hydrocarbons contain a set of covalently bound atoms with specific characteristics: • A delocalized conjugated π system, with the common arrangement of alternating single and double bonds
Aliphatics Alkanes Cycloalkanes Alkenes Alkynes
Alkanes IUPAC ending is …ane
Alkanes • Saturated hydrocarbons. • Are hydrocarbons which contain only single bonds. • All alkanes are insoluble in water, but dissolve in organic solvents. • Density, viscosity, melting point & boiling points increase as the molecular weight/size of the hydrocarbon increases.
Alkanes • Contain single covalent bonds. • Have the same structural formula: CnH2n+2 • All carbons have single bonds therefore the molecular geometry is tetrahedral.
Alkanes • The names of alkanes start with the name of the alkane but end with the suffix –ane.
Alkanes • Each atom in an alkane uses all its 4 valence electrons in forming single bonds with other atoms. • Alkyl groups may be used as substituents for hydrogens.
Alkanes • Alkyl groups form the branches of straight chain hydrocarbons. • Can have more than one alkyl group for hydrogens. • For multiple substituents of the same type, use the following prefixes: • di- • tri- • tetra- • penta- • hexa-
Alkanes • Other functional groups can be used as substituents. • More than one substituent requires a prefix. • Any hydrogen can be substituted by: Halogens Alcohols Amines Nitriles Thiols Aldehydes Ketones
Alkanes • Any carbon can be substituted by: • Carboxylic Acids • Esters • Amides • Thioesters • Addition of other atoms: • Ethers • Thioethers • Disulfides
Cycloalkanes the prefix cyclo- and the ending …ane
Cycloalkanes • Saturated hydrocarbons. • Form one or more rings fused together. • A single carbon in a ring may have two hydrogen atoms. • Are insoluble in water, but dissolve in organic solvents. • Have higher boiling points, melting points, and densities than alkanes.
Cycloalkanes • All have the same general formula: CnH2n • The carbon atoms in cycloalkanes are sp3hybridized. • Each atom in a cycloalkane uses all its 4 valence electrons in forming covalent bonds with other atoms.
Cycloalkanes • Can have more than one alkyl group to make straight chains. • For multiple alkyl groups of the same type, use prefixes.
Cycloalkanes • Many functional groups can be used as substituents. • More than one substituent requires a prefix. • Any hydrogen or carbon atom can be substituted by:
Cycloalkanes • The names follow those of the alkanes with the prefix cyclo- .
Alkenes IUPAC ending is …ene
Alkenes • Also known as olefins. • Are unsaturated hydrocarbons and are generally very reactive. • Are insoluble in water, but dissolve in organic solvents. • Ethene, propene and buteneare gases at room temperature. The remaining are liquids. • Boiling points increases with molecular mass (chain length). The higher the molecular mass, the higher the boiling point.
Alkenes • Are hydrocarbons which contain one or more double bonds. • Double bonds are: • Have the same structural formula: CnH2n
Alkenes • The main centers are the carbons of the double bond. • The geometry of each carbon in the center is trigonal planar. • This portion of the molecule is flat, with bond angles of 120 degrees.
Alkenes All the alkenes with 4 or more carbon atoms in them show structural isomerism.
Alkenes • The carbon-carbon double bond does not rotate. • Substituents groups on the molecule are locked on either one side of the molecule or opposite each other.
Alkenes • The names of alkenes start with the name of the alkane but end with the suffix –ene. • For alkenes above propene, the position of the double bond must be specified in the name.
Alkenes • Can have more than one alkyl group to form branches. • For more than one alkyl group, use prefixes.
Alkenes • Many functional groups can be used as substituents. • More than one substituent requires a prefix. • Any hydrogen or carbon atom can be substituted by:
Alkenes • For multiple double bonds, use the following prefixes: • di- • tri- • tetra- • penta- • hexa-
Alkenes • A diene is a hydrocarbon chain that has two double bonds that may or may not be adjacent to each other.
Alkynes IUPAC ending is …yne
Alkynes • Also known as acetylenes. • Are unsaturated hydrocarbons and are generally very reactive. • Are insoluble in water; but quite soluble in organic solvents of low polarity (e.g. ligroin, ether, benzene, carbon tetrachloride, etc.). • Alkynes of four or fewer carbon atoms are gases. The rest are liquids. • Their boiling points increase with increasing number of carbons.
Alkynes • Are hydrocarbons which contain one or more triple bonds. • Triple bonds are: • Have the same structural formula: CnH2n-2
Alkynes • The main centers are the carbons of the triple bond. • The geometry of the center is linear. • This portion of the molecule is linear, with bond angles of 180 degrees.
Alkynes All the alkynes with 4 or more carbon atoms in them show structural isomerism.
Alkynes • The names of alkynes start with the name of the alkane but end with the suffix –yne. • For alkynes above propyne, the position of the triple bond must be specified in the name.
Alkynes • Many functional groups can be used as substituents. • Only one substituent is allowed. • Any hydrogen or carbon atom can be substituted by:
Alkynes • For multiple double bonds, use the following prefixes: • di- • tri- • tetra- • penta- • hexa-
Aromatics Structures that meet Huckel’s Rule
Aromatics • Coplanar structures, with all the contributing atoms in the same plane. • Are arranged in one or more rings. • Benzene rings are not a common motif. • The three general requirements for a compound to be aromatic are: • The compound must be cyclic. • Each element within the ring must have a p-orbital that is perpendicularto the ring, hence the molecule is planar. • The compound must follow Hückel'sRule.