Periodic Table
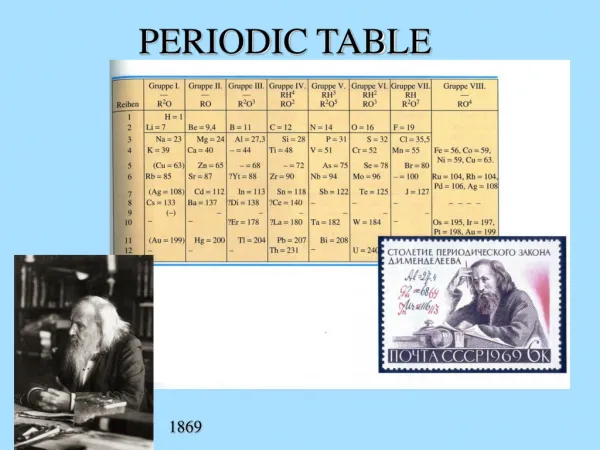
Periodic Table. Periodic Law : When all elements are arranged in order of increasing atomic numbers, elements with similar chemical properties will occur are regular (periodic) intervals.

Periodic Table
E N D
Presentation Transcript
Periodic Table Periodic Law: When all elements are arranged in order of increasing atomic numbers, elements with similar chemical properties will occur are regular (periodic) intervals. In modern periodic table, elements with similar chemical properties are arranged in vertical columns called groups or families. The horizontal rows in the periodic table are called periods.
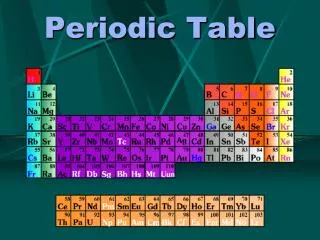
Groups and Periods Periodic table group or family • Traditionally designated by a Roman numeral and a letter (either A or B) at the top of the column. • Designated only by a number from 1 to 18 in a modern but as yet not universally-used designation. Periodic table Period • Periods are numbered numerically from top to bottom of the periodic table. In a modern table, elements 58-71 and 90-103 are not placed in their correct periods, but are located below the main table.
Electronic arrangements in Atoms Rutherford’s atom Solar system model Niels Bohr proposed that the electron in hydrogen atom could occupy orbits only at specific distances from the nucleus. In other words, the electron moved in any one of a series of circular orbits around the nucleus. Electron can change orbits only by absorbing or releasing energy, with higher energy orbit located farther from the nucleus.
Quantum Mechanical model of atom According to the quantum mechanical model of electron behavior, the precise paths of electrons moving around the nucleus cannot be determined accurately. Instead of circular orbits, the location and energy of electrons moving around the nucleus is specified using the three terms shell, subshell and orbital. Locate your position on map 1 Locate your position on map 2 Locate your position on map 3
Shell The location of electrons in a shell is indicated by assigning a number n to the shell and all electrons located in the shell. The value of n is a whole number- 1, 2, 3, 4 etc. The higher the n value, • the higher is the energy of the shell • the higher is the contained electrons • the greater is the distance from the nucleus
Subshell Each shell is made up of one or more subshells that are designated by a letter from the group s, p, d, or f. The number of the shell to which a subshell belongs is combined with the letter of the subshell to clearly identify subshells. For example, a p subshell located in the third shell (n = 3) would be disignated as a 3p subshell. The number of subshells located in a shell is the same as the number of the shell. Thus, shell number 3 (n = 3) contains three subshells, designated 3s, 3p, and 3d. Electrons located in a subshell are often identified by using the same designation as the subshell they occupy. Thus electrons in a 3d subshell are called 3d electrons.
Atomic Orbitals The description of the location and energy of an electron moving around a nucleus is completed in the quantum mechanical model by specifying an atomic orbital in which the electron is located. Each subshell consists of one or more atomic orbitals, which are specific volumes of space around the nucleus in which electrons move. Atomic orbitals are designated by the same number and letter used to designate the subshell to which they belong. Thus, an s orbital located in a 2s subshell would be called a 2s orbital.
Atomic Orbitals All s subshells consist of a single s orbital. All p subshells consist of three p orbitals. All d subshells consist of five d orbitals. All f subshells consist of seven f orbitals. According to the quantum mechanical model, all types of atomic orbitals can contain a maximum of two electrons. Thus, a single d orbital can contain a maximum of 2 electrons, and a d subshell that contains seven d orbitals can contain a maximum of 14 electrons.
Atomic Orbitals Atomic Orbital shapes The shape of the orbital should not be confused to mean that the electrons move around the orbital shape surface. It only determines the probability to find the electron in a specific location.
Energy of electrons The energy of electrons around a nucleus depends on two factors. • Electron energy increases with increasing n value. Thus an electron in the third shell (n = 3) has more energy than an electron in the first shell (n = 1). • For equal n values but different orbitals, the energy of electrons in orbitals increases in the order s, p, d and f. Thus, a 4p electron has more energy than a 4s electron. Example 3.3
Shell model and chemical properties The outermost occupied shell (with the highest n value) is called as the valence shell. Atoms with the same number of electrons in the valence shell have similar chemical properties. Example 3.4
Electronic Configurations Electronic configurations give details of the arrangements of electrons in atoms. The notation used to represent electronic configurations is 1s22s22p6 etc., where the occupied subshells are indicated by their identifying number and letter such as 2s, and the number of electrons in the subshell is indicated by the superscript on the letter. Thus, in the example above, the 2s2 notation indicates that the 2s subshell contains two electrons. Electrons will fill subshells in the order of increasing energy of the subshells. Thus, a 1s subshell will fill before a 2s subshell. The order of subshell filling must obey Hund's rule and the Pauli exclusion principle.
Electronic Configurations Hund’s rule- • According to Hund's rule, electrons will not join other electrons in an orbital of a subshell if an empty orbital of the same energy is available in the subshell. • Thus, the second electron entering a p subshell will go into an empty p orbital of the subshell rather than into the orbital that already contains an electron. The Pauli exclusion principle- • Electrons behave as if they spin on an axis. • According to the Pauli exclusion principle, only electrons spinning in opposite directions can occupy the same orbital within a subshell. Combined- Electrons will pair with other electrons in an orbital only if there is no empty orbital of the same energy available and if there is one electron with opposite spin already in the orbital.
Electronic Configurations Example 3.6 a, b
Relative energies and electron-filling order for shells and subshells Some low energy subshells of a specific shell have energies lower than the upper subshell of a preceding shell. 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f, 6d, 7p
Electronic Configurations Electronic configuration for: An atom that contains 17 electrons (Example 3.6) An atom that contains 23 protons (Learning Check 3.6)
Electronic Configurations Electronic Configuration gives information about the shells, subhsells and orbitals. But are cumbersome. Hence, use Noble Gas configuration: An electronic configuration in which the last eight electrons occupy and fill the s and p subshells of the highest-occupied shell. Noble gas configurations can be used to write electronic configurations in an abbreviated form in which the noble gas symbol enclosed in brackets is used to represent all electrons found in the noble gas configuration. Sodium: [Ne]3s1. The symbol [Ne] represents the electronic configuration of neon, 1s22s22p6. Magnesium: An atom that contains 17 electrons An atom that contains 23 protons [Ne]3s2. The symbol [Ne] represents the electronic configuration of neon, 1s22s22p6.
Classification according to Distinguishing Electrons The distinguishing electron is the last electron listed in the electronic configuration of the element.
Representative, Transition, Inner-transition, Noble Gases Noble Gases: Groups VIII A filled s and p subshells (exception He) Representative elements: s and p areas (other than VIII A) Transition elements: d area Inner-transition elements: f area
Metals, Nonmetals & Metalloids Metals- elements in the left two-thirds High thermal/electrical conductivity, ductility, malleability, metallic luster. Metalloids- elements narrow diagonal band between metals and nonmetals Some characteristics of each Nonmetals- elements in the right one-third Brittle, powdery solids or gases
Periodic Trends • Trends in Metallic Properties • Elements in the same period of the periodic table become less metallic and more nonmetallic from left to right across the period. • Elements in the same group of the periodic table become more metallic and less nonmetallic from top to bottom down the group. • Trends in the size of atoms • For representative elements in the same period, atomic size decreases from left to right in the period • For representative elements in the same group, atomic size increases from top to bottom down the group.
Metallic and size of atoms trends for representative elements
Periodic Trends Ionization Energy of an element is the energy required to remove an electron from an atom of the element in the gaseous state. This results in formation of a charged species called ion. First ionization energy is the energy to remove the first electron from a neutral atom. • Trends in Ionization Energy • For representative elements in the same period, the general trend is an increase from left to right across the period. • For representative elements in the same group, the general trend is a decrease from top to bottom down the group.
Colors of elements Form a group with 3 other students having different color cards Green, Blue, Yellow, Pink Choose an element belonging to your color Describe the element’s properties in terms of: Metal/Nonmetal/Metalloid Size of atom (if possible) Representative, Transition, Inner-transition, Noble gas Write an abbreviated electronic configuration (inner transition folks may skip this)