Balloon Analogy for the Mutual Repulsion of Electron Groups
480 likes | 1.24k Vues
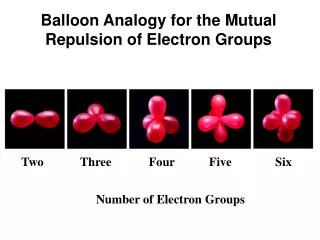
Balloon Analogy for the Mutual Repulsion of Electron Groups. Two. Three Four Five Six. Number of Electron Groups. AX 2 Geometry - Linear. . . . . Molecular Geometry = Linear Arrangement. . . Cl. Be. Cl. BeCl 2. 180 0.

Balloon Analogy for the Mutual Repulsion of Electron Groups
E N D
Presentation Transcript
Balloon Analogy for the MutualRepulsion of Electron Groups Two Three Four Five Six Number of Electron Groups
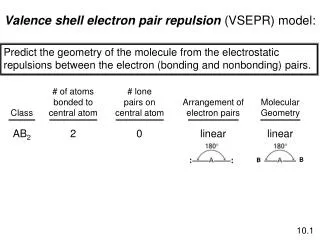
AX2 Geometry - Linear .. .. .. .. Molecular Geometry = Linear Arrangement .. .. Cl Be Cl BeCl2 1800 Gaseous beryllium chloride is an example of a molecule in which the central atom - Be does not have an octet of electrons, and is electron deficient. Other alkaline earth elements also have the same valence electron configuration, and the same geometry for molecules of this type. Therefore this geometry is common to group II elements. .. .. .. .. O C O CO2 1800 Carbon dioxide also has the same geometry, and is a linear molecule, but in this case, the bonds between the carbon and oxygens are double bonds.
The Two Molecular Shapes of the Trigonal Planar Electron-Group Arrangement
S O O AX3 Geometry - Trigonal Planar .. .. .. .. All of the boron Family(IIIA) elements have the same geometry. Trigonal Planar ! .. .. F F BF3 B Boron Trifluoride 1200 .. .. .. F AX2E SO2 .. - .. .. .. .. O .. .. NO3- 1200 .. .. N .. .. .. .. O O The AX2E molecules have a pair of Electrons where the third atom would appear in the space around the central atom, in the trigonal planar geometry. 1200 Nitrate Anion
The Three Molecular Shapes of the Tetrahedral Electron-Group Arrangement
107.30 .. N N H H H AX4 Geometry - Tetrahedral H Methane H 109.50 CH4 H C H C H H H H All molecules or ions with four electron groups around a central atom adopt the tetrahedral arrangement H H 109.50 109.50 + H+ H Ammonia is in a tetrahedral shape, but it has only an electron pair in one location, so the smaller angle! all angles are the same! Ammonium Ion H
The Four Molecular Shapes of the Trigonal Bipyramidal Electron- Group Arrangement
AX5 Geometry - Trigonal Bipyramidal .. .. .. .. .. .. F I .. .. .. 86.20 .. .. .. I 1800 Br .. F .. .. .. .. .. I .. F .. AX3E2 - BrF3 .. AX2E3 - I3- .. .. .. Cl .. .. Cl .. .. P Cl .. AX5 - PCl5 .. .. .. .. .. Cl .. Cl
The Three Molecular Shapes of the Octahedral Electron-Group Arrangement
AX6 Geometry - Octahedral .. .. .. .. .. .. .. .. .. .. F .. F .. .. .. F .. F .. .. .. F F .. .. .. .. S I .. .. .. .. .. .. F F .. .. F F .. .. .. .. F AX5E Iodine Pentafluoride AX6 Sulfur Hexafluoride .. .. .. .. .. .. F .. F .. Xe .. .. .. .. .. F .. F Xenon Tetrafluoride Square planar shape
Using VSEPR Theory to Determine Molecular Shape 1) Write the Lewis structure from the molecular formula to see the relative placement of atoms and the number of electron groups. 2) Assign an electron-group arrangement by counting all electron groups around the central atom, bonding plus nonbonding. 3) Predict the ideal bond angle from the electron-group arrangement and the direction of any deviation caused by the lone pairs or double bonds. 4) Draw and name the molecular shape by counting bonding groups and non-bonding groups separately.
The sp3d2 Hybrid Orbitals in SF6 Sulfur Hexafluoride -- SF6
Restricted Rotation of -Bonded Molecules A) Cis - 1,2 dichloroethylene B) trans - 1,2 dichloroethylene
Postulating the Hybrid Orbitals in a Molecule Problem: Describe how mixing of atomic orbitals on the central atoms leads to the hybrid orbitals in the following: a) Methyl amine, CH3NH2b) Xenon tetrafluoride, XeF4 Plan: From the Lewis structure and molecular shape, we know the number and arrangement of electron groups around the central atoms, from which we postulate the type of hybrid orbitals involved. Then we write the partial orbital diagram for each central atom before and after the orbitals are hybridized.
Postulating the Hybrid Orbitals in a Molecule Problem: Describe how mixing of atomic orbitals on the central atoms leads to the hybrid orbitals in the following: a) Methyl amine, CH3NH2b) Xenon tetrafluoride, XeF4 Plan: From the Lewis structure and molecular shape, we know the number and arrangement of electron groups around the central atoms, from which we postulate the type of hybrid orbitals involved. Then we write the partial orbital diagram for each central atom before and after the orbitals are hybridized. Solution: a) For CH3NH2: The shape is tetrahedral around the C and N atoms. Therefore, each central atom is sp3hybridized. The carbon atom has four half-filled sp3 orbitals: 2s 2p sp3 Isolated Carbon Atom Hybridized Carbon Atom
The N atom has three half-filled sp3orbitals and one filled with a lone pair. 2s sp3 2p .. H C N H H H H
b) The Xenon atom has filled 5 s and 5 p orbitals with the 5 d orbitals empty. Isolated Xe atom 5 d 5 s 5 p Hybridized Xe atom: sp3d2 5 d
.. .. b) continued:For XeF4. for Xenon, normally it has a full octet of electrons,which would mean an octahedral geometry, so to make the compound, two pairs must be broken up, and bonds made to the four fluorine atoms. If the two lone pairs are on the equatorial positions, they will be at 900 to each other, whereas if the two polar positions are chosen, the two electron groups will be 1800 from each other. Thereby minimizing the repulsion between the two electron groups. F F F F 1800 Xe Xe F F F F Square planar