Investigating Chiral Recognition in Electrokinetic Chromatography via NMR Spectroscopy
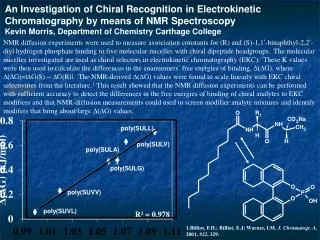
This study investigates the chiral recognition of (R) and (S)-1,1’-binaphthyl-2,2’-diyl hydrogen phosphate using NMR diffusion experiments to measure association constants with five molecular micelles featuring chiral dipeptide headgroups. These micelles act as chiral selectors in electrokinetic chromatography (EKC). The binding free energy differences (D(DG)) were calculated and shown to scale linearly with EKC chiral selectivities found in literature, suggesting that NMR diffusion experiments can effectively identify chiral analyte binding behaviors to EKC modifiers.

Investigating Chiral Recognition in Electrokinetic Chromatography via NMR Spectroscopy
E N D
Presentation Transcript
An Investigation of Chiral Recognition in Electrokinetic Chromatography by means of NMR Spectroscopy Kevin Morris, Department of Chemistry Carthage College NMR diffusion experiments were used to measure association constants for (R) and (S)-1,1’-binaphthyl-2,2’-diyl hydrogen phosphate binding to five molecular micelles with chiral dipeptide headgroups. The molecular micelles investigated are used as chiral selectors in electrokinetic chromatography (EKC). These K values were then used to calculate the differences in the enantiomers’ free energies of binding, D(DG), where D(DG)=|DG(S) –DG(R)|. The NMR-derived D(DG) values were found to scale linearly with EKC chiral selectivities from the literature.1 This result showed that the NMR diffusion experiments can be performed with sufficient accuracy to detect the differences in the free energies of binding of chiral analytes to EKC modifiers and that NMR-diffusion measurements could used to screen modifier:analyte mixtures and identify modifers that bring about large D(DG) values. poly(SULL) poly(SULV) poly(SULA) poly(SULG) poly(SUVV) poly(SUVL) 1.Billiot, F.H.; Billiot, E.J; Warner, I.M. J. Chromatogr. A, 2001, 922, 329.