The Haber Process: Ammonia Production and Fritz Haber's Controversial Legacy
Explore the Haber Process used for ammonia production, its importance in creating fertilizer and explosives, and Fritz Haber's controversial Nobel Prize-winning legacy. Learn how Haber maximized ammonia yield and the dark history of his involvement with chemical warfare during WWI. Discover the impact of Nazi persecution on his life.

The Haber Process: Ammonia Production and Fritz Haber's Controversial Legacy
E N D
Presentation Transcript
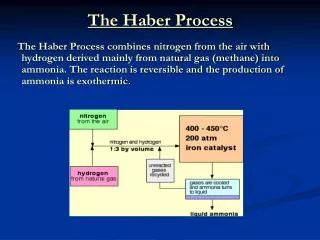
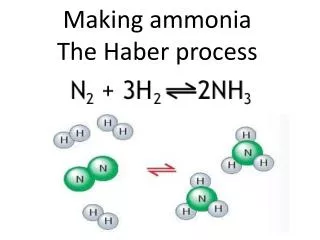
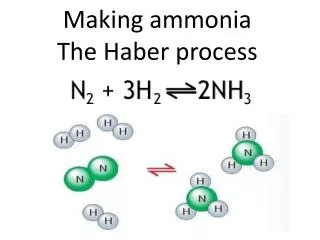
Haber Process Fritz Haber The Haber Process is used to make ammonia, NH3: 3 H2(g) + N2 (g) ↔ 2 NH3(g) + heat Ammonia ranks in the top five of all chemicals in terms of total mass produced. It is used to make fertilizer and explosives (e.g.- TNT).
Even though the Haber process has a very large Kc (5 X 108), the reaction rate is virtually zero at room temperature and pressure. How did Haber maximize the yield rate of production AND yield of ammonia? http://www.freezeray.com/flashFiles/ammoniaConditions.htm
Fritz Haber was awarded the Nobel Prize in Chemistry in 1918, but the award was contested because Haber made it possible for Germany to use mustard gas as a weapon during WWI. Fritz Haber explaining the benefits of poison gas.
Haber was forced to leave Germany in 1933 because of Nazi persecution of persons of Jewish ethnicity.