Haber Process
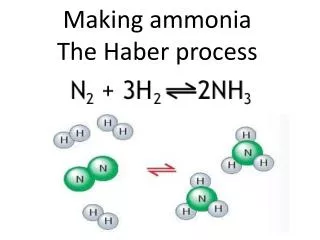
Haber Process. Making of ammonia from its elements. Uses of Ammonia. Fertilisers , fibers, plastics, nitric acid (explosives), household cleaners, detergents. N 2 and H 2. Ammonia. Chemical monitoring. Equilibrium. To produce ammonia:

Haber Process
E N D
Presentation Transcript
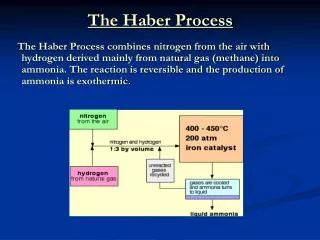
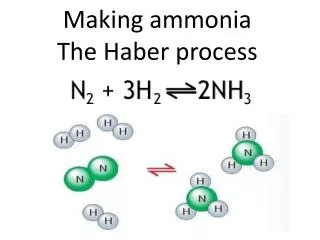
Haber Process Making of ammonia from its elements
Uses of Ammonia Fertilisers, fibers, plastics, nitric acid (explosives), household cleaners, detergents
N2 and H2 Ammonia Chemical monitoring
Equilibrium To produce ammonia: Increase pressure (it will tend to decrease the number of moles of gas) Decrease temperature (it will tend to increase temperature)
Increasing pressure increases the frequency of successful collisions • Compromise: Construction of strong pipes to maintain high pressures is not economical. Pressure needs to be lessened Pressure
Lower temperature produces the highest yield of ammonia (~90%) but at a very slow rate (months!) as there is insufficient kinetic energy for a collision • Compromise: Use slightly higher temperatures to yield smaller amounts quickly. Temperature
Catalysts speed the forward and reverse reaction. • It does not change the position of equilibrium • Compromise: A catalyst makes it possible to use lower temperatures Catalyst
Conditions: Increase reactants (but must maintain 1:3 ratio) Pressure: 25 MPa (25,000 Pa) Temperature: 400oC Magnetite Catalyst Product is removed
Yield of ammonia: ~15–20% per cycle. After 5–6 cycles about 98% of the reactants are converted to ammonia.
Why Monitor? Efficiency and safety