Absorption and Scattering
Absorption and Scattering. Peng Xi Changhui Li 北京大学工学院 生物医学工程系. 2011/09/09. The Propagation of Light. The processes of transmission, reflection, and refraction are macroscopic manifestations of scattering occurring on a submicroscopic level.


Absorption and Scattering
E N D
Presentation Transcript
Absorption and Scattering Peng Xi Changhui Li 北京大学工学院 生物医学工程系 2011/09/09
The Propagation of Light The processes of transmission, reflection, and refraction are macroscopic manifestations of scattering occurring on a submicroscopic level. http://faculty.qu.edu.qa%2Fmalrabban%2FOptics%2FOptics_08Propagation.ppt
Elastic Scattering • In elastic scattering, the energy of the incident photonis conserved and its propagating direction is changed by thepotential of the target.
Rayleigh Scattering When a photon penetrates into a medium composed of particles whose sizes are much smaller than the wavelength of the incident photon, the scattering process is elastic and is called Rayleigh scattering. In this scattering process, the energy (and therefore the wavelength) of the incident photon is conserved and only its direction is changed. In this case, the scattering intensity is proportional to the fourth power of the reciprocal wavelength of the incident photon. The scattering of electromagnetic radiation by particles with dimensions much smaller than the wavelength of the radiation, resulting in angular separation of colors and responsible for the reddish color of sunset and the blue of the sky.
Assume The intensity of the scattered light Example 4.1 Establish the dependence of the percentage of light scattered in Rayleigh scattering. Let is the incident amplitude, is the scattered amplitude at a distance r from the scatterer. V is the volume of the scatterer.
Must be unitless, and K must has units of ( Length )2
The Transmission of Light Through Dense Media Little or no light ends up scattered laterally or backwards in a dense homogeneous medium. This makes sense from the perspective of conservation of energy– we can’t have constructive interference in every direction. Interference produces a redistribution of energy, out of the regions where it’s destructive into the regions where it’s constructive.
Constructive vs. destructive interference;Coherent vs. incoherent interference Constructive interference(coherent) Waves that combine in phase add up to relatively high irradiance. = Waves that combine 180° out of phase cancel out and yield zero irradiance. Destructive interference(coherent) = Waves that combine with lots of different phases nearly cancel out and yield very low irradiance. Incoherent addition =
Scattering from molecules and small particles A plane wave impinging on a molecule or particle scatters into a spherical wave. Huygens’ Principle says that waves propagate as if each point on a wave-front emits a spherical wave (whether or not there’s a molecule or particle involved). Scattering from an individual molecule or particle is weak, but many such scatterings can add up—especially if interference is coherent and constructive.
The Phases of the wavelets at P differ greatly The Transmission of Light Through Dense Media
Waves using complex amplitudes • The resulting "complex amplitude" is: As written, this entire field is complex!
Complex numbers simplify optics! Adding waves of the same frequency, but different initial phase, yields a wave of the same frequency. This isn't so obvious using trigonometric functions, but it's easy with complex exponentials: where all initial phases are lumped into E1, E2, and E3.
time time time Adding complex amplitudes When two waves add together with the same complex exponentials, we add the complex amplitudes, E0 + E0'. Constructive interference: Destructive interference: "Quadrature phase" ±90° interference: 1.0 0.2 1.2 1.0 -0.2 0.8 1.0 -0.2i 1-0.2i + + + = = = Slower phase velocity Laser Absorption
Incident light + Emitted light = Transmitted light Light excites atoms, which emit light that adds (or subtracts) with the input light. When light of frequency w excites an atom with resonant frequency w0: Electric field at atom Electron cloud Emitted field On resonance (w = w0) An excited atom vibrates at the frequency of the light that excited it and re-emits the energy as light of that frequency. The crucial issue is the relative phase of the incident light and this re-emitted light. For example, if these two waves are ~180° out of phase, the beam will be attenuated. We call this absorption.
Light excites atoms, which then emit more light. Incident light Emitted light + Transmitted light = The interaction of light and matter Electric field at atom Electron cloud Emitted electric field On resonance (the light frequency is the same as that of the atom) The crucial issue is the relative phase of the incident light and this re-emitted light. If these two waves are ~180° out of phase, destructive interference occurs, and the beam will be attenuated—absorption. If they’re ~±90° out of phase: the speed of light changes—refraction.
Electric field at atom Electron cloud Emitted field The relative phase of emitted light with respect to the input light depends on the frequency. Weak emission.90° out of phase. Below resonance w << w0 Strong emission.180° out of phase. On resonancew = w0 The emitted light is 90° phase-shifted with respect to the atom’s motion. Weak emission.-90° out of phase. Above resonance w >> w0
Refractive index and Absorption coefficient w0 Frequency, w
Dispersed beam Input white beam Prism Variation of the refractive index with wavelength (dispersion) causes the beautiful prismatic effects we know and love. Prisms disperse white light into its various colors.
Light Scattering When light encounters matter, matter not only re-emits light in the forward direction (leading to absorption and refractive index), but it also re-emits light in all other directions.This is called scattering. Light scattering is everywhere. All molecules scatter light. Surfaces scatter light. Scattering causes milk and clouds to be white and water to be blue. It is the basis of nearly all optical phenomena. Scattering can be coherent or incoherent.
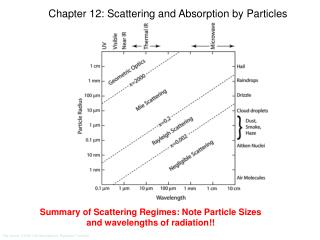
Particle size/wavelength ~0 ~1 Large Rayleigh-Gans Scattering Large ~1.5 ~0 Refractive index Mie Scattering Rayleigh Scattering Geometrical optics Totally reflecting objects There are many regimes of particle scattering, depending on the particle size, the light wavelength, and the refractive index. You can read an entire book on the subject: Light scattering regimes Air Rainbow
The mathematics of scattering If the phases aren’t random, we add the fields: Coherent Etotal = E1 + E2 + … + En Itotal = I1 + I2 + … + In I1, I2, … In are the irradiances of the various beamlets. They’re all positive real numbers and add. Ei Ej* are cross terms, which have the phase factors: exp[i(qi-qj)]. When the q’s are not random, they don’t cancel out! If the phases are random, we add the irradiances: Incoherent
The Biological Origin of Light Scattering College of Engineering, Peking University生物医学光学II
Absorption • The absorption coefficient (μa) is defined as the probability of photon absorption in a medium per unit path length (strictly speaking, per unit infinitesimal path length). • The absorption coefficient can be considered as the total cross-sectional area for absorption per unit volume.
Beer-Lambert Law • The Beer-Lambert law (or Beer's law) is the linear relationship between absorbance and concentration of an absorbing species. The general Beer-Lambert law is usually written as: A = a(λ) * b * c where A is the measured absorbance, a(λ) is a wavelength-dependent absorptivity coefficient, b is the path length, and c is the analyte concentration. • When working in concentration units of molarity, the Beer-Lambert law is written as: A = ε* b * c where εis the wavelength-dependent molar absorptivity coefficient with units of M-1 cm-1. http://elchem.kaist.ac.kr/vt/chem-ed/spec/beerslaw.htm
Ballistic photons • Snake photons • Diffused photons
Trajectories of Optical Photons in Biological Tissue Reflectometry Laser beam Tissue 1 mm Photoacoustics
Spectra of Major Biological Absorbers Near IR window: ~700 nm ~1 µm penetration 2.95 µm
Melanin: Eumelanin: A black-to-dark-brown insoluble material found in human black hair and in the retina of the eye. Pheomelanin: A yellow-to-reddish-brown alkali-soluble material found in red hair and red feathers. Polymers ~10 nm in diameter Studding the inner walls of melanosomes (~1 micron diameter organelle) Volume fraction of melanosome in epidermis: Light skinned Caucasions: 1-3% Dark pigmented Africans: 18-43% Spectrum of Melanosome http://omlc.ogi.edu/spectra
Spectrum of Fat (Lipids) http://omlc.ogi.edu/spectra
Spectrum of Methylene Blue Dye:Contrast Agent Used in Sentinel Lymph Node Mapping C16H18ClN3S, MW 319.85. Also called Swiss blue. One gram dissolves in about 25 ml of water, or in 65 ml alcohol. Peak absorption at 668 and 609 nm. --- Merck Index http://omlc.ogi.edu/spectra
Spectrum of Indocyanine Green (ICG):Cardiac Output and Hepatic Function Measurements C43H47N2O6S2Na, Molecular weight 775. A tricarbocyanine type of dye with infrared absorbing properties; peak absorption at about 800nm. Has little or no absorption in the visible. It is used in infrared photography and in the preparation of Wratten filters. It is also used as a diagnostic aid for blood volume determination, cardiac output, or hepatic function. --- Merck Index http://omlc.ogi.edu/spectra
Molar Extinction Spectra of Hemoglobin [nm] 259.93 339.54 390.01 422.05 452.36 500.11 529.24 545.26 570.18 584.09 796.80 Isosbestic point
Scattering The scattering coefficient (mus) is defined as the probability of photon scattering in a medium per unit path length (strictly speaking, per unit infinitesimal path length). The scattering coefficient can be considered as the total cross-sectional area for scattering per unit volume.
Extinction Extinction = absorption + scattering Molar extinction coefficient = extinction coefficient per Molar concentration per length Molar = moles/L
The Distribution of Scattered Radiation (Phase Function) Geometrical Optics Rayleigh Resonance Incoming light direction Adapted from http://hyperphysics.phy-astr.gsu.edu/hbase/atmos/blusky.html W. P. Arnott, AAAR tutorial, Sept. 2007 38
Example of a morning when the Mexico City Plume Goes South to Popocatepetl Volcano. near forward scattering by particles sca = 30 degrees r << r ~ r >> 39 W. P. Arnott, AAAR tutorial, Sept. 2007
Basic properties n [–]: index of refraction; e.g., 1.37 µa [cm–1]: absorption coefficient; e.g., 0.1 µs [cm–1]: scattering coefficient; e.g., 100 g [–]: scattering anisotropy, <cosq>; e.g., 0.9 Derived properties µt [cm–1]: total interaction (extinction) coefficient, µa + µs lt [cm]: mean free path, 1/ µt; e.g., 0.1 mm µs’ [cm–1]: reduced scattering coefficient, µs(1 – g) µt’ [cm–1]: transport interaction coefficient, µa + µs’ lt’ [cm]: transport mean free path, 1/ µt’; e.g., 1 mm µeff [cm–1]: effective attenuation coefficient, (3µa µt’)1/2 δ [cm]: penetration depth, 1/(3µa µt’)1/2; e.g., 5 mm Optical Properties of Biological Tissue
CFM: Confocal microscopy 2PM: Two-photon microscopy OCT: Optical coherence tomography DOT: Diffuse optical tomography PAT: Photoacoustic tomography lt’: Optical transport mean free path ~ 1 mm (mean free path ~ 0.1 mm) δ: Effective penetration depth Major Challenge in Optical Tomography: High Resolution Beyond the Quasiballistic (~1-mm Depth) Regime Laser 1 mm CFM & 2PM Soft limit* ~ lt’ OCT DOT, PAT Hard limit ~ 10δ ~ 5-7 cm (20,000X or 43 dBone-way attenuation) * Applied Optics 38, 4951 (1999). Simulation software MCML available on the web
Scattering • Elastic • Rayleigh scattering • Mie scattering • Inelastic • Raman scattering • The difference in energy generates a vibrational excitation in the molecule • Brillouin scattering • The difference in energy generates acoustic phonons.
Summary • Absorption: Beer’s law • Biological • Scattering-Elastic: • Rayleigh scattering: 1/λ4 • Mie scattering: weak relative to wavelength