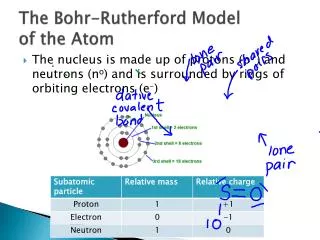
element 44 = atomic # 44 44 protons 44 electrons
120 likes | 849 Vues
66. How many protons and electrons are contained in an atom of element 44?. element 44 = atomic # 44 44 protons 44 electrons. 67. A carbon atom has a mass number of 12 and an atomic number of 6. How many neutrons does it have?. mass number (p + n) 12 atomic number (p) - 6

element 44 = atomic # 44 44 protons 44 electrons
E N D
Presentation Transcript
66. How many protons and electrons are contained in an atom of element 44? element 44 = atomic # 44 44 protons 44 electrons
67. A carbon atom has a mass number of 12 and an atomic number of 6. How many neutrons does it have? • mass number (p + n) 12 • atomic number (p)- 6 • = # of neutrons 6 n
68. An isotope of mercury has 80 protons and 120 neutrons. What is the mass number of the isotope? protons 80 + neutrons + 120 = Mass number 200
69. An isotope of xenon has an atomic number of 54 and contains 77 neutrons. What is the xenon isotope’s mass number? protons 54 + neutrons + 77 = Mass number 131
70. Is an atom has 18 electrons, how many protons does it have? 18 protons
73. How many electrons, protons, and neutrons are contained in each? electrons protons neutrons 13255 Cs 55 55 77 b) 5927 Co 27 27 32 c) 16369 Tm 69 69 94 d) 7030 Zn 30 30 40
74. How many electrons, protons, and neutrons are contained in each? electrons protons neutrons Gallium -69 31 31 38 b) fluorine-23 9 9 14 c) titanium-48 22 22 26 d) tantalum-181 73 73 108
76. Ga, which has atomic mass of 69.723 amu, has two naturally occurring isotopes, Ga-69 and Ga-71. Which isotope occurs in greater abundance? Explain. Ga- 69 must be more abundant because the atomic mass of gallium is closer to the mass of Ga-69 than the mass of Ga-71
77. Silver has two isotopes 10747 Ag, which has a mass of 106.905 amu and a percent abundance of 52.00% and 10947 Ag , which has a mass of 108.905 amu and a percent abundance of 48.00%. What is the atomic mass of silver? 106.905 x .5200 = 55.59 108.905 x .4800 = 52.27 107.86 amu
78. Data for chromium’s four naturally occurring isotopes are provided in the table. Calculate the atomic mass (amu). = 2.17 = 43.52 = 5.03 = 1.27 51.99 amu