Molybdenum
Molybdenum. 1900 – discovered in plant ashes 1930 - azotobacter (N2 fixer) growth requirement for Mo 1940 – established as essential micronutrient for plants 1950 - [Mo] affected rat XO activity; [Mo]~ [flavin] Mo associated with aldehyde oxidase in rabbit livers

Molybdenum
E N D
Presentation Transcript
Molybdenum 1900 – discovered in plant ashes 1930 - azotobacter (N2 fixer) growth requirement for Mo 1940 – established as essential micronutrient for plants 1950 - [Mo] affected rat XO activity; [Mo]~ [flavin] Mo associated with aldehyde oxidase in rabbit livers 1960 - Mo crucial to N-cycle MoS The only 4d metal essential to Life
Table 1. The Oxomolybdenum Enzymes (R. Hille, Chem. Rev. 1996) The Xanthine Oxidase Family (LMoOS-Possessing Enzymes) enzyme source subunits cofactora xanthine oxidase cow’s milkb R2 MPT xanthine dehydrogenase chicken liverc R2 MPT rat liverd R2 MPT Micrococcus lactyliticuse Drosophila melanogasterf R2 MPT Chlamydomonas reinhardtiig humanh R2 (MPT) aldehyde oxidase rabbit liveri R2 (MPT) humanj R2 (MPT) cowk R2 (MPT) aldehyde oxidoreductase (dehydrogenase) Desulfovibrio gigasl R2 MCD Acetobacter polyoxogenesm formate dehydrogenase Alcaligenes eutrophusn Râçä Methylosinus trichosporumo R2â2ç2ä2 CO dehydrogenase (oxidoreductase) Pseudomonas carboxydovoransp R2â2ç2 Pseudomonas carboxydoflavaq R2â2ç2 MCD Oligotropha carboxidovoransr R2â2ç2 MCD quinoline-2-oxidoreductase Pseudomonas putida s R2â2ç2 MCD Rhodococcus sp. B1t R2â2ç2 MCD Comamonas testosteroni 63u R2â2ç2 MCD isoquinoline 1-oxidoreductase Pseudomonas diminutav Râ MCD quinoline-4-carboxylate-2-oxidoreductase Agrobacterium sp. 1Bw R2â2ç2 MCD quinaldine-4-oxidoreductase Arthrobacter sp.x R2â2ç2 MCD quinaldic acid 4-oxidoreductase Serratia marcescensy Pseudomonas sp. AK-2z Râ nicotinic acid hydroxylase (dehydrogenase) Clostridium barkeriaa,ab R2 Bacillus niaciniac R2â2ç2 Arthrobacter oxidansad Râç 6-hydroxynicotinate hydroxylase Bacillus niaciniac Râç nicotine dehydrogenase Arthrobacter oxidansad Râç Arthrobacter nicotinovoransae picolinate hydroxylase Arthrobacter picolinophilusaf R2â2ç2 MCD (2R)-hydroxycarboxylate oxidoreductase Proteus vulgarisag The Sulfite Oxidase Family (LMoO2-Possessing Enzymes) sulfite oxidase bovine liverah R2 MPT chicken liverai R2 MPT rat liveraj R2 MPT humanak R2 MPT Thiobacillus novellusal R nitrate reductase (assimilatory) Neurospora crassaam R2 MPT spinachan R2 MPT Chlorella vulgarisao R4 MPT The DMSO Reductase Family (L2MoX-Possessing Enzymes) DMSO reductase Rhodobacter sphaeroidesap R MGD Rhodobacter capsulatus aq R MGD Escherichia coli ar Râç MGD biotin-S-oxide reductase Escherichia colias trimethylamine-N-oxide reductase Escherichia coliat R2 nitrate reductase (dissimilatory) Escherichia coli (NarGHI)au Râç MGD Escherichia coli (NarZYV)av Râç MGD Escherichia coli (FdoGHI)aw Râç MGD Paracoccus denitrificans (NapABCD)ax Râçä Haloferax volcaniiay formate dehydrogenase Escherichia coli (FdhF)az R MGD Escherichia coli (FdnGHI)ba Râç MGD Escherichia coli (FdoGHI)bb Râç MGD Methanobacterium formicicumbc (XdS) Râç MGD Wollinella succinogenesbd Râç polysulfide reductase Wolinella succinogenesbe Râç MGD arsenite oxidase Alcaligenes faecalisbf R MCD formylmethanofuran dehydrogenase) Râçä(ú) MGD, MAD, MHD Methanosarcina barkeribh MGD Unclassified Molybdenum-Containing Enzymes pyridoxal oxidase Drosophila melanogaster chlorate reductase Proteus mirabilis tetrathionite reductase Proteus mirabilis xanthine dehydrogenase Clostridium sp pyruvate:ferredoxin oxidoreductase Archaeoglobus fulgidus pyrogallol transhydroxylase Pelobacter acidigallici 2-furoyl-CoA dehydrogenase Pseudomonas putida
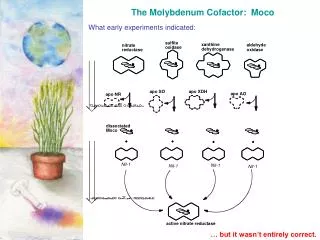
Locating the Molybdenum Cofactor All mammals require molybdenum enzymes Sulfite Oxidase SO32- + H2O SO42- + 2H+ + 2e- All plants require the molybdenum enzyme Nitrate Reductase NO3- + 2H+ + 2e- NO2- + H2O Mo(4+) + X-O + 2H+ Mo(6+) + X + H2O
NO2- N2 Nitrate reductase nitrogenase Nitrogen- cycle NO3- NH3 polysulfide reductase SH2 CH4 Sx Carbon- cycle Sulfur- cycle CO2 CH3CO2- (CH3)2S (CH3)2SO DMSO reductase
Table 1. The Oxomolybdenum Enzymes (R. Hille, Chem. Rev. 1996) The Xanthine Oxidase Family (LMoOS-type) enzyme source cofactor xanthine oxidase cow’s milk MPT xanthine dehydrogenase chicken liver MPT rat liver MPT Drosophila melanogaster MPT human MPT) aldehyde oxidase rabbit liver MPT human MPT cow MPT aldehyde oxidoreductase (dehydrogenase) Desulfovibrio gigas MCD formate dehydrogenase Alcaligenes eutrophus CO dehydrogenase (oxidoreductase) Pseudomonas carboxydovorans quinoline-2-oxidoreductase Pseudomonas putida MCD isoquinoline 1-oxidoreductase Pseudomonas diminuta MCD quinoline-4-carboxylate-2-oxidoreductase Agrobacterium sp MCD quinaldine-4-oxidoreductase Arthrobacter sp. MCD quinaldic acid 4-oxidoreductase Serratia marcescens Pseudomonas sp. AK nicotinic acid hydroxylase (dehydrogenase) Clostridium barkeriaa, 6-hydroxynicotinate hydroxylase Bacillus niaciniac nicotine dehydrogenase Arthrobacter oxidansa The Sulfite Oxidase Family (LMoO2-type) enzyme source cofactor sulfite oxidase bovine liver MPT chicken liver MPT rat liver MPT human MPT Thiobacillus novellus nitrate reductase (assimilatory) Neurospora crassa MPT spinach MPT Chlorella vulgari MPT The DMSO Reductase Family (L2MoX-type) DMSO reductase Rhodobacter sphaeroides MGD biotin-S-oxide reductase Escherichia colia trimethylamine-N-oxide reductase Escherichia colia nitrate reductase (dissimilatory) Escherichia coli (NarGHI) MGD formate dehydrogenase Escherichia coli (FdhF) MGD polysulfide reductase Wolinella succinogene MGD arsenite oxidase Alcaligenes faecalisb MCD formylmethanofuran dehydrogenase) MGD, MAD, MHD Unclassified Molybdenum-Containing Enzymes pyridoxal oxidase Drosophila melanogaster chlorate reductase Proteus mirabilis tetrathionite reductase Proteus mirabilis xanthine dehydrogenase Clostridium sp. pyruvate:ferredoxin oxidoreductase Archaeoglobus fulgidus pyrogallol transhydroxylase Pelobacter acidigallici 2-furoyl-CoA dehydrogenase Pseudomonas putida
The Generic Molybdenum Cofactor MAD MGD MCD MPT There’s not only one, but a family of Moco’s
dithiolene pterin Why molybdopterin? Mo
Table 1. The Oxomolybdenum Enzymes (R. Hille, Chem. Rev. 1996) The Xanthine Oxidase Family (LMoOS-type) The Sulfite Oxidase Family (LMoO2-type) The DMSO Reductase Family (L2MoX-type) Used to be classed with XO b/c structural similarity. Unique Mo enzyme with a 2nd metal, Cu
Table 1. The Oxomolybdenum Enzymes (R. Hille, Chem. Rev. 1996) The Xanthine Oxidase Family (LMoOS-type) Xanthine Oxidase/Dehydrogenase - in mammalian milk but >50% de-Mo - in liver: purine metabolism (gout) - implicated in heart damage (ROS form’n) The Sulfite Oxidase Family (LMoO2-type) Nitrate Reductase - in all plants - assimilatory form (plants, fungi, algae) Sulfite Oxidase - most critical for humans - detoxification of sulfite - also has role in S-metabolism, from Cys Met SO32- SO42- The DMSO Reductase Family (L2MoX-type) - all bacterial or from archea - many in respiration using specific e- acceptors - recall DMSOR in cloud formation …. - simplest Mo-enzymes: w/o other cofactors
Table 1. The Oxomolybdenum Enzymes (R. Hille, Chem. Rev. 1996) The Xanthine Oxidase Family (LMoOS-type) - in mammalian milk but >50% de-Mo - in liver: purine metabolism (gout) - implicated in heart damage (ROS form’n) The Sulfite Oxidase Family (LMoO2-type) Nitrate Reductase - in all plants - assimilatory form (plants, fungi, algae) NO3- + 2H+ + 2e- NO2- + H2O Sulfite Oxidase - most critical for humans - detoxification of sulfite - also has role in S-metabolism, from Cys Met SO32- SO42- SO32- + H2O SO42- + 2H+ + 2e- The DMSO Reductase Family (L2MoX-type) - all bacterial or from archea - many in respiration using specific e- acceptors - recall DMSOR in cloud formation …. - simplest Mo-enzymes: w/o other cofactors
Root nodules formed by Rhizobium bacteria living symbiotically through nitrogen fixation.
Tobacco plants (Arabidopsis thaliana) Nitrate Nitrite Proteins nitrate nitrite Healthy (wild type) Sick (mutant) All plants require Assimilatory Nitrate Reductase
Introduction Repairing the Molybdenum Cofactor Baby Z Cured of Rare Disease in 3 Days Orphan Drug Treatment Used Only on Mice to Get Hearing Before FDA By SUSAN DONALDSON JAMES, Nov. 9, 2009 (Southern Health/AFP/Getty Images) Baby Z had a one in a million chance of developing a rare metabolic disorder called molybdenum cofactor deficiency and zero chance of avoiding the inevitable death sentence that comes with it. The Australian girl had a seemingly normal birth in May 2008 but, within hours, she began having multiple seizures -- as many as 10 an hour -- as sulfite build-up began to poison her brain. With the clock ticking, doctors who treated Baby Z gained approval from the hospital's ethics board and a family court to use the experimental treatment. The drug -- cPMP, a precurser molecule made from E. coli bacteria -- was airlifted on ice from the lab of German professor Guenter Schwarz and, within three days, it worked. University of Arizona, Tucson, October 2010 Worldwide, there are only about 50 cases of molybdenum cofactor, or sulfite oxidase deficiency, mostly in Europe and in the United States, according to the National Institutes of Health. Molybdenum, like other organic metals, is essential for the human body. Its cofactor is a small, complicated molecule that acts as a carrier to help the metal interact with proteins and enzymes so they can function properly. When the cofactor is missing, toxic sulfite builds and begins to cause degeneration of neurons on the brain and eventually death. "This was the first time I ever saw this," said Dr. Alex Veldman, the Monash neonatologist who headed up Baby Z's treatment. "It's very funny, now I am regarded a world specialist but I can tell you that before last May, I couldn't even spell it."
MRI of brain of deceased baby with Sulfite Oxidase Deficiency MRI of healthy brain
The baby died because this reaction didn’t happen: SO32- + H2O ---> SO42- + 2H+ + 2e- Sulfite Sulfate S4+ S6+ The baby has a genetic defect in the enzyme that catalyzes this reaction. Babies with this genetic disease die within hours. The enzyme is Sulfite Oxidase. You have it in your liver.
Review the terms: Oxidation SO32- + H2O ---> SO42- + 2H+ + 2e- half reactionS4+ in Sulfite S6+ in Sulfate Reduction Mo6+ + 2e- ---> Mo4+ half reaction Net redox reaction SO32- + H2O + Mo6+ ---> SO42- + 2H+ + Mo4+ Is this reaction spontaneous?
Caroline Kisker Würzburg, Germany Protein crystallographer X-ray structure of chicken liver Sulfite Oxidase Kisker, Enemark
Similar cycles can be devised for Sulfite Oxidase and Nitrate Reductase SO32- + H2O ---> SO42- + 2H+ + 2e- Sulfite Sulfate S4+ S6+ NO3- + 2H+ + 2e- ---> NO2- + H2O Nitrate Nitrite N5+ N3+
DMS CH3SO3- cloud nucleation sites hn, photo-oxidation DMS (CH3)2S(CH2)2CO2- from algae DMSO Reductase DMSO Environmental impact of DMSOR and the smell of the ocean
DMSOR Structure: Controversy #2 The first Mo enzyme X-ray structure: DMSO Reductase Doug Rees, 1996 Doug Rees, Cal Tech Protein crystallographer Group Meeting Bryn Mawr College, October 2010 • SURPRISE!!!! • 2 molydopterin ligands! • nucleoside termini on pterin • very long Mo-S bonds
DMSOR Structure: Controversy #2 The first look at molybdopterin was on a tungsten enzyme! Hyperthermophilic TungstonEnzyme, Aldehyde Ferredoxin Oxidoreductase Doug Rees et al., Science,1995 Group Meeting Bryn Mawr College, October 2010 • SURPRISE!!!! • not the molydopterin ligand! • is that pyran ring actually right???
DMSOR Structure: Controversy #2 Will the real active site structure in DMSO Reductase please stand up? Group Meeting Bryn Mawr College, October 2010 (S J N Burgmayer, in Progress in Inorganic Chemistry, 2004)
Hermann Schindelin, Würzburg, Germany Protein crystallographer DMSOR Structure: Controversy #2 1.3 Å X-ray Structure in DMSO Reductase (Schindelin) Group Meeting Bryn Mawr College, October 2010 Inactive form Active form What does it mean? There are 2 superimposed structures. (only one is inactive!)