Patient Management: Renal System
2.41k likes | 3.12k Vues
Dr. Aidah Abu Elsoud Alkaissi An-Najah National University Faculty of Nursing. Patient Management: Renal System. Renal Function. Renal function may be replaced by a process called dialysis, which is a life-maintaining therapy used in acute and chronic renal failure.

Patient Management: Renal System
E N D
Presentation Transcript
Dr. Aidah Abu Elsoud Alkaissi An-Najah National University Faculty of Nursing Patient Management:Renal System
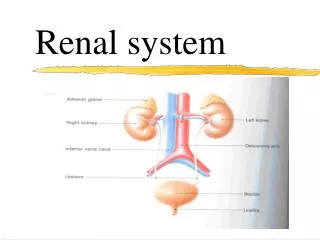
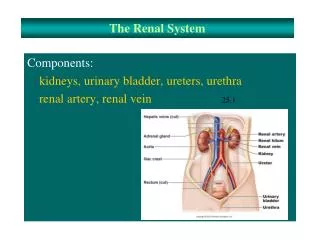
Renal Function Renal function may be replaced by a process called dialysis, which is a life-maintaining therapy used in acute and chronic renal failure. Critical care nurses may encounter patients suffering from the effects of acute renal failure or patients already on some form of chronic dialysis who subsequently become critically ill. Critical care nurses must be familiar with various dialysis therapies to help care for patients with complex illnesses.
Renal Function The three most common forms of renal replacement therapy: hemodialysis, continuous renal replacement therapies [CRRTs], common fluid and electrolyte imbalances experienced by critically ill patients.
PHYSIOLOGY All forms of dialysis make use of the principles of osmosis and diffusion to remove waste products and excess fluid from the blood. A semipermeable membrane is placed between the blood and a specially formulated solution called dialysate.
PHYSIOLOGY Dissolved substances, such as urea and creatinine,diffuse across the membrane from an area of greater concentration (blood) to an area of lesser concentration (dialysate). Water molecules move across the membrane by osmosis to the solution that contains fewer water molecules
PHYSIOLOGY Dialysate is formulated with varying concentrations of dextrose or sodium to produce an osmotic gradient, thereby pulling excess water from the circulatory system. This process of fluid moving across a semipermeable membrane in relation to forces created by osmotic and hydrostatic pressures is called ultrafiltration.
EXTRACORPOREAL THERAPIES Hemodialysis and the CRRTs use an extracorporeal (outside the body) circuit. Therefore, they require access to the patient’s circulation and anticoagulation of the circuit.
Access to Circulation The three most common methods used to access a patient’s circulation are through a vascular catheter, an arteriovenousfistula, or a synthetic vascular graft. Patients who suddenly need hemodialysis or CRRT have a venous catheter, whereas patients already receiving chronic hemodialysis probably have either an arteriovenous fistula or a synthetic vascular graft.
Nursing interventions for the Patient With Dialysis Vascular AccessDual-Lumen Venous Catheter ■ Verify central line catheter placement radiographically before use. ■ Do not inject IV fluids or medication into the catheter. Both lumens of the catheter usually are filled with concentrated heparin. ■ Do not unclamp the catheter unless preparing for dialysis therapy. This can cause blood to fill the lumen and clot. ■ Maintain sterile technique in handling vascular access. ■ Observe catheter exit site for signs of inflammation or access limb.
Arteriovenous Fistula or Graft ■ Do not take blood pressure or draw blood from the hand that have the fistula ■ Listen for bruit (reflecting turbulence of flow) and palpate for thrill q8h. ■ Make sure there is no tight clothing or restraints on the access limb. ■ Check access patency more frequently when patients are hypotensive. Hypotension can predispose to clotting. ■ In the event of postdialysis bleeding from the needle site, apply just enough pressure to stop the flow of blood and hold until bleeding stops. Do not occlude the vessel.
VENOUS CATHETERS acutely ill patients who need hemodialysis, continuous venovenous hemofiltration (CVVH), or continuous venovenous hemofiltration with dialysis (CVVH/D). Venous catheters are also used for hemodialysis when there is no other means of access to the circulation. Veins commonly used are the femoral, internal jugular, or subclavian.
VENOUS CATHETERS The site chosen depends on the patient’s anatomy and vein accessibility and the physician’s experience and preference.
Dual-lumen venous catheters also are used temporarily for patients on acute dialysis who are critically ill or patients on chronic dialysis who are waiting for a more permanent access to mature. Tunneled dual-lumen central venous catheters are often used as a permanent means of access in patients in whom all other means of entry into the circulatory system have been exhausted.
The tunneled catheter has an implantable Dacron cuff around which tissue grows and acts as a barrier against infection. If possible, the catheter should be placed in the right or left internal jugular vein because catheters placed in the subclavian vein can cause stenosis. The stenosis can cause increased venous pressure and edema that may thwart (To prevent the occurrence) future efforts to create an arteriovenous fistula or place a graft.
Whenever venous catheters are used, care must be taken to avoid accidental slippage and dislodgement during hemodialysis. Femoral catheters are usually secured with sutures as well as with tape to the leg, whereas central venous catheters in the upper body are sutured to the skin. The length of time catheters are left in place depends on catheter function and institution policy. In general, central venous catheters may be used for up to 3 to 4 weeks.
permanent internal jugular vein catheters often function for many months before problems force their removal. Catheters left in place between dialysis treatments usually are filled with a concentrated heparin–saline solution after dialysis and plugged to prevent clotting.
These catheters should never be used for any purpose other than hemodialysis without first checking with dialysis unit personnel. Cleansing and dressing of the insertion site are the same as with other central lines. If the catheters are removed at the end of dialysis, pressure is applied to the puncture sites until complete clotting occurs. The site is checked for several hours thereafter so that any recurrent bleeding can be detected. Removal of the more permanent tunneled catheter requires use of local anesthetic at the exit site and careful dissection around the Dacron cuff to free it from the attached subcutaneous tissue.
Catheter patency must be maintained. Thrombolytics may be used to dissolve clots in venous catheters. Thrombolytics are enzymes derived from streptococcal bacteria that are capable of activating the fibrinolytic system and dissolving intravascular thrombi. These agents can help preserve vascular access and reduce the need for surgery or catheter reinsertion. However, their use is associated with inherent risks and side effects, including local pain, bleeding, and an allergic response.
In the early days of dialysis, vascular access was created at every treatment by cannulating an artery to remove blood from the body and a vein to return dialyzed blood to the patient. The lines carrying blood to the dialyzer were called arterial lines, and the lines returning blood to the body were called venous lines. The two lumens of the venous catheter used in dialysis are still designated as arterial and venous.
The arterial lumen is longer than the venous lumen so it can catch blood flowing by and allow it to be pumped out of the body. Blood is returned upstream from the arterial lumen to avoid pulling out the blood that has just been dialyzed and returned to the body. The lumens are distinguished by the presence of colored clamps: red on the arterial lumen and blue on the venous lumen.
ARTERIOVENOUS FISTULAS The arteriovenous fistula technique was developed in 1966 in an effort to provide long-term access for hemodialysis. To create the arteriovenous fistula, a surgeon anastomoses an artery and a vein, creating a fistula or artificial opening between them
Arterial blood flowing into the venous system results in a marked dilation of the vein, which can then be punctured easily with a 15- or 16-gauge dialysis fistula needle. Two venipunctures are made at the time of dialysis: one for blood outflow and one for blood return.
After the arteriovenous fistula incision has healed, the site is cleansed by normal bathing or showering. To avoid scar formation, excessive bleeding, or hematoma of the arteriovenous fistula, care is taken to avoid traumatic venipuncture, excessive manipulation of the needles, and repeated use of the same site fo venipuncture.
Adequate pressure must be put on the puncture sites after the needles are removed. In addition, blood pressure measurements and venipunctures should not be performed on the arm with the fistula. Most arteriovenous fistulas are developed and ready to use 1 to 3 months after surgery. After initial healing has occurred, patients are taught to exercise the arm to assist in vessel maturation. They also are encouraged to become familiar with the quality of the “thrill” felt at the site of anastomosis so that they can report any change in its presence or strength.
A loud, swishing sound termed a bruit indicates a functioning fistula. Although arteriovenous fistulas usually have a long life, complications may occur. These include thrombosis, aneurysm or pseudoaneurysm, or arterial insufficiency causing a “steal syndrome.” This syndrome occurs when shunting of blood from the artery to the vein produces ischemia of the hand, causing pain or coldness in the hand. Surgical intervention can remedy all of these problems and restore adequate fistula flow.
Caring for an Arteriovenous Fistula ■ Wash the fistula site with antibacterial soap each day and always before dialysis. ■ Refrain from picking the scab (A crust discharged from and covering a healing wound) that forms after completion of dialysis therapy. ■ Check for redness, feeling of excess warmth, or the beginning of a pimple on any area of access. ■ Ask the dialysis care team to rotate needles at the time of dialysis treatment. ■ Check blood flow several times each day by feeling for a pulse or thrill. If this is not felt, or if there is a change, call your health care provider or dialysis center.
Caring for an Arteriovenous Fistula ■Refrain from wearing tight clothes or jewelry on the access arm. Also avoid carrying anything heavy or doing anything that will put pressure on the access site. ■ Avoid sleeping with your head on the arm where the access site is located. ■ Remind caregivers and staff not to use a blood pressure cuff on, or draw blood from, the arm where the access site is located. ■ Apply only gentle pressure to the access site after the needle is removed. Too much pressure stops flow of blood to the access site.
SYNTHETIC GRAFTS The synthetic graft is made from polytetrafluoroethylene (PTFE), a material manufactured from an expanded, highly porous (Full of or having pores) form of Teflon. The graft is anastomosed between an artery and a vein and is used in the same manner as an arteriovenous fistula
For many patients whose own vessels are not adequate for fistula formation, PTFE grafts are extremely valuable. PTFE segments are also used to patch areas of arteriovenous grafts or fistulas that have stenosed or developed areas of aneurysm. It is best to avoid venipuncture in new PTFE grafts for 2 to 4 weeks while the patient’s tissue grows into the graft.
When tissue growth progresses satisfactorily, the graft has an endothelium and wall composition similar to the patient’s own vessels. The procedures for preventing complications in grafts are the same as those used for arteriovenous fistulas. However, certain complications are seen more frequently with grafts than with fistulas, including thrombosis, infection, aneurysm formation, and stenosis at the site of anastomosis.
Anticoagulation • Blood in the extracorporeal system, such as the dialyzer • and blood lines, clots rapidly unless some method of anticoagulation is used. • Heparin is the drug of choice because it is simple to administer, increases clotting time rapidly, is monitored easily, and may be reversed with protamine. Trisodium citrate 4% may also be used for CRRT. • This agent chelates (Chemical compounds that act as water "softening" agents. "Hard" minerals (such as calcium) are attracted to and can neutralize the surfactants in a detergent. Chelates do not remove or exchange hard minerals in water. Rather, chelate molecules surround and cover (sequester) those minerals calcium), thereby inactivating the clotting • cascade.
Specific heparinization procedures vary, but the primary goal of all methods is to prevent clotting in the dialyzer with the least amount of anticoagulation. Two methods commonly used are intermittent and constant infusion.
SYSTEMIC ANTICOAGULATION Typically, the circuit is initially primed with a dose of heparin, followed by smaller intermittent doses of anticoagulation or heparin administered at a constant rate by an infusion pump. This results in systemic anticoagulation,in which the clotting times of the patient and the dialyzer essentially are the same.
Definitive guidelines are difficult to provide because methods and dialyzer requirements vary. The normal clotting time of 6 to 10 minutes may be increased to 30 to 60 minutes. The effect of heparin usually is monitored by the activated clotting time, prothrombin time (PT), or partial thromboplastin time (PTT).
The patient’s need for heparinization and an appropriate beginning heparin dose should be assessed routinely before dialysis, especially in the critically ill patient who may be actively bleeding or at risk for bleeding. The patient’s platelet count, serum calcium level, and results of coagulation studies are valuable in assessing current function of the clotting process. Often little or no heparin can be used when the patient has serious alterations in one or more factors needed for effective clotting.
REGIONAL ANTICOAGULATION Systemic heparinization does not usually present a risk unless the patient has overt bleeding (e.g., gastrointestinal bleeding, epistaxis, or hemoptysis), is 3 to 7 days postsurgery, or has uremic pericarditis. In these situations, other methods to prevent clotting of the extracorporeal system can be used
One method is regional heparinization, in which the patient’s clotting time is kept normal while the clotting time of the dialyzer is increased. This is accomplished by infusing heparin at a constant rate into the dialyzer and simultaneously neutralizing its effects with protamine sulfate before the blood returns to the patient.
Like systemic heparinization, regional heparinization has no associated standard heparin–protamine ratio. Frequent monitoring of the clotting times is the best way to achieve effective regional heparinization. Because of the rebound phenomenon that has occurred after regional heparinization, low-dose heparinization may be used, even in the presence of overt bleeding. With this method, minimal heparin doses are used throughout dialysis. Although some clotting may take place in the dialyzer, the small blood loss is preferable to the risk of profound bleeding.
Bleeding problems occasionally occur because of accidental heparin overdose. This may be caused by infusion pump malfunction or an error in setting the delivery rate. Because of the hazards, heparin delivery must be monitored carefully and frequently
. Another way to prevent dialyzer clotting and reduce the risk of bleeding due to heparin is to infuse a small initial heparin dose (e.g., 250 U) and use frequent normal saline flushes of the extracorporeal system, or use saline flushes alone.
Some dialysis centers perform regional citrate anticoagulation in which citrate is infused into the system before the dialyzer binds calcium, obstructing the normal clotting pathway. The citrate–calcium complex is then cleared from the blood by the dialyzer, and the anticoagulant effect is reversed by infusing calcium chloride before the blood returns to the patient. The patient’s sodium levels may rise because the citrate is administered in the form of sodium citrate. Citrate has a higher pH, and therefore patients may also become metabolically alkalotic.
INTERMITTENT HEMODIALYSIS In hemodialysis, water and excess waste products are removed from the blood as it is pumped by the dialysis machine through an extracorporeal circuit into a device called a dialyzer, or artificial kidney. The blood is in one compartment, and the dialysate is in another compartment. There, the blood flows through a semipermeable membrane.
The semipermeable membrane is a thin, porous sheet made of cellulose or a synthetic material. The pore size of the membrane permits diffusion of low–molecular-weight substances such as urea, creatinine, and uric acid. In addition, water molecules are small and move freely through the membrane, but most plasma proteins, bacteria, and blood cells are too large to pass through the pores of the membrane. The difference in the concentration of the substances in the two compartments is called the concentration gradient.
The blood, which contains waste products such as urea and creatinine, flows into the blood compartment of the dialyzer, where it comes into contact with the dialysate,which contains no urea or creatinine. A maximum gradient is established so that these substances move from the blood to the dialysate. These waste products fall to more normal levels as the blood passes through the dialyzer repeatedly at a rate ranging from 200 to 400 mL/minute over 2 to 4 hours
Excess water is removed by a pressure differential created between the blood and fluid compartments. This pressure differential is aided by the action of the dialyzer pump and usually consists of positive pressure in the blood path and negative pressure in the dialysate compartment. This is the process of ultrafiltration.
Hemodialysis: ■ Removes byproducts of protein metabolism, such as urea, creatinine, and uric acid ■ Removes excess water ■ Maintains or restores the body buffer system ■ Maintains or restores the level of electrolytes in the body
Indications for Hemodialysis Hemodialysis is indicated in chronic renal failure and for complications of acute renal failure. These include uremia, fluid overload, acidosis, hyperkalemia, and drug overdose. Table 30-1 compares hemodialysis, CRRT, and peritoneal dialysis.
Contraindications to Hemodialysis Hemodialysis may be contraindicated in patients with coagulopathies because the extracorporeal circuit needs to be heparinized. Hemodialysis may also be difficult to perform in patients who have extremely low cardiac output or who are sensitive to abrupt changes in volume status. For these critically ill patients, CRRT may be the optimal choice. In addition, intermittent hemodialysis may not keep up with the metabolic needs of a highly catabolic patient. In this case, CRRT would probably be chosen. Patients treated chronically for renal failure may be given the choice to undergo hemodialysis or peritoneal dialysis.
Equipment DIALYZERS Dialyzers are designed to provide a parallel path through which blood and dialysate flow and to have a maximal membrane surface area between the two. Dialyzers vary in size, physical structure, and type of membrane used to construct the blood compartment. All these factors determine the potential efficiency of the dialyzer, which refers to its ability to remove water (ultrafiltration) and waste products (clearance).
The hollow-fiber dialyzer is the most commonly used configuration. In this design, the blood path flows through hollow fibers composed of semipermeable membrane, and the dialysate path is encased in a rigid plastic tube. Dialysate surrounds each hollow fiber. This provides a large surface area to cleanse the blood. Blood and dialysate flow in opposite directions from each other (countercurrent flow); as blood travels through the dialyzer, it is constantly exposed to a fresh flow of dialysate. This countercurrent flow maintains the concentration gradient between the two compartments and provides the most efficient dialysis