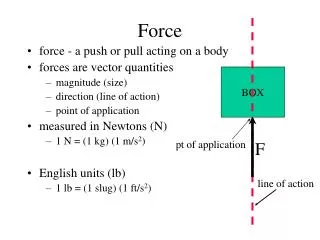
Thermal Force
Thermal Force. Unit 1.4. Objectives. Define thermal energy Name the property of a body that determines its temperature. Given Celsius or Fahrenheit temperature and the formula for conversion, find the equivalent temperatures. Explain the difference between heat and thermal energy

Thermal Force
E N D
Presentation Transcript
Thermal Force Unit 1.4
Objectives • Define thermal energy • Name the property of a body that determines its temperature. • Given Celsius or Fahrenheit temperature and the formula for conversion, find the equivalent temperatures. • Explain the difference between heat and thermal energy • Explain the relationship between heat transferred to an object and the change in the object’s temperature • Use specific heat, heat of fusion, and heat of vaporization to solve problems involving heat transfer
Terms (We will study energy in the next unit) • Energy – the ability to do work. • Potential energy – the stored or build up of energy; the capacity/ “potential” to do work. • Kinetic energy – energy of motion. • Thermal motion – the random motion of vibration of an object’s atoms and molecules. • Thermal energy – the total energy of the thermal motion of all particles that make up an object
Temperature • Temperature is the “hotness” property of a body; it is the average kinetic energy of the random motion of the atoms and molecules in a body. • Heat flows from regions of higher temperature to regions of lower temperature. • Measured in degrees Fahrenheit or degrees Celsius. • Thermal Energy is measured in Joules, Calories and BTU’s
Measuring Temperature • Thermometer – uses expansion and contraction of a liquid usually colored alcohol or mercury. • Celsius scale – developed by Anders Celsius and based on the properties of water. (0° = freezing; 100° = boiling) • Fahrenheit scale - used in the US (32° = freezing and 212° = boiling)
Temperature scales 5 TC = (TF – 32) 9 9 TF = TC + 32 5
Heat • Conduction – thermal energy flows from the hotter object to the cooler object as kinetic energy is transferred when particles collide. • Heat is the energy that flows from one object to another because of temperature difference. • Heat flow depends on temperature difference. • As the temperature difference increases so does the heat transfer rate. (no temp. diff = no heat transfer) • Temperature difference is the prime mover in thermal systems.
Analogies to Prime Movers • Mechanical – Force • Fluid – Pressure • Electrical – Voltage • Thermal – Temperature Difference
Specific Heat • The specific heat (C) of a substance is the amount of energy that must be added to raise the temperature of a unit mass of the substance one unit of temperature. energy cal Specific heat of water = 1 g • C° Unit of temp. mass
Change of State • States of matter – Solid, Liquid, Gas, and Plasma.
Changes in State • Freezing Point – Temperature at which a substance changes from a liquid to a solid. • Boiling Point – Temperature at which a liquid changes to a gas. • Heat of Fusion (Hf)– the amount of energy required to change a melt a solid. • Heat of Vaporization (Hv) – the amount of energy required to vaporize a liquid.
Changes of State • The amount of heat (Q) needed to melt a solid of mass (m) is • Q = m Hf • The mount of heat (Q) needed to vaporize a liquid of mass (m) is • Q = m Hv
Summary • The thermal energy of a body is the total kinetic energy of motion of all the particles that make up the body. • The temperature of a body is determined by the average kinetic energy of the particles that make up the body. • A thermometer measures temperature in degrees Celsius or degrees Fahrenheit. • Heat is the energy that flows from one body to another because of a temperature difference. • Whenever two bodies are brought together, heat flows from the body with higher temperature to the body with the lower temperature. • The amount of heat transferred to an object varies linearly with the object’s temperature change, as long as there is no change of state: Q=mcT • If heat is transferred to a substance and it changes state, its temperature does not change.
Measurements • One calorie is the amount of thermal energy that must be added to water to change the temperature of 1 gram of water by 1 degree Celsius. • One Btu is the amount of thermal energy that must be added to water to change the temperature of 1 pound of water by 1 degree Fahrenheit.
Heat Transfer Object’s mass Temperature Change Q = m C T Heat transferred Specific heat