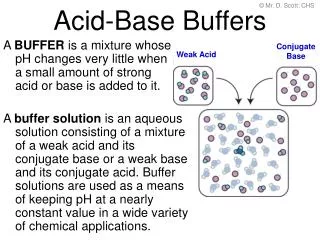
A solution that contains a weak acid/conjugate base mixture or weak base/conjugate acid mixture.
110 likes | 498 Vues
Buffer Solutions. A solution that contains a weak acid/conjugate base mixture or weak base/conjugate acid mixture. Resists changes in pH when a moderate amount of acid or base is added to it Made two ways: Weak acid and one of its salts OR Weak base and one of its salts.

A solution that contains a weak acid/conjugate base mixture or weak base/conjugate acid mixture.
E N D
Presentation Transcript
Buffer Solutions • A solution that contains a weak acid/conjugate base mixture or weak base/conjugate acid mixture. • Resists changes in pH when a moderate amount of acid or base is added to it • Made two ways: Weak acid and one of its salts OR Weak base and one of its salts
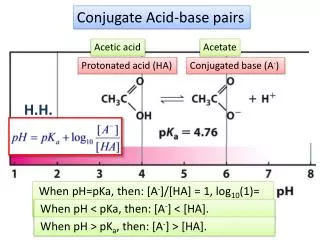
Characteristics of a buffer solution: • pH • buffer capacity • amount of acid or base that can be added before considerable change to pH • depends on concentration of acid/conjugate base or base/conjugate acid in buffer solution • if more concentrated, resists changes to pH more than a buffer that is more dilute • Blood has a buffer solution with hydrogen carbonate ions and carbonate ions, which is important to life
How does a buffer solution resist changes in pH with the addition of an acid or a base? Le Châtelier’s Principle
Acid-Base Titration Curves Acid-Base Titration • Common analytical procedure used to determine the concentration of a reactant, using pH, acids, bases and indicators • Equivalence point • point in a titration when the acid and base that are present completely react with each other • middle of steep rise that occurs in titration curve • If you know volume of both and concentration of one, you can find the concentration of the other.
Endpoint • Point in titration where indicator changes colour • Pick an indicator that changes colour close to the equivalence point
Strong Acid-Strong Base Titration Curve • pH = 7 at equivalence point • Use indicators like phenolphthalein, methyl red, bromocresol green
Weak Acid-Strong Base Titration Curve pH >7 at equivalence point Use indicators like phenolphthalein
Weak Base-Strong Acid Titration Curve • pH <7 at equivalence point • Use indicators like methyl red, bromocresol green
References • McGraw-Hill Ryerson Chemistry 12











![pH = -log [H + ] . Strong Acid . Strong base . Weak Acid. Weak Base.](https://cdn3.slideserve.com/6574453/slide1-dt.jpg)





