Chapter 21 Enzymes and Vitamins
160 likes | 735 Vues
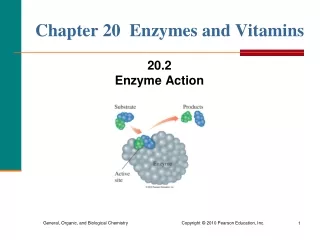
Chapter 21 Enzymes and Vitamins. 21.5 Enzyme Inhibition 21.6 Regulation of Enzyme Activity 21.7 Enzyme Cofactors. Enzyme Inhibition. Inhibitors : Are molecules that cause a loss of catalytic activity. Prevent substrates from fitting into the active sites. E + S ES E + P

Chapter 21 Enzymes and Vitamins
E N D
Presentation Transcript
Chapter 21 Enzymes and Vitamins 21.5 Enzyme Inhibition 21.6 Regulation of Enzyme Activity 21.7 Enzyme Cofactors
Enzyme Inhibition Inhibitors: • Are molecules that cause a loss of catalytic activity. • Prevent substrates from fitting into the active sites. E + S ES E + P E + I EI no P
Reversible Competitive Inhibition A competitive inhibitor: • Has a structure like the substrate. • Competes with the substrate for the active site. • Has its effect reversed by increasing substrate concentration.
Noncompetitive Inhibition A noncompetitive inhibitor: • Has a structure different than the substrate. • Distorts the shape of the enzyme, which alters the shape of the active site. • Prevents the binding of the substrate. • Cannot have its effect reversed by adding more substrate.
Malonate and Succinate Dehydrogenase Malonate: • Is a competitive inhibitor of succinate dehydrogenase. • Has a structure that is similar to succinate. • Inhibition is reversed by adding succinate.
Irreversible Inhibition • Loss of all enzymatic activity • Toxic substance (irreversible inhibitor) forms a covalent bond with an amino acid in the active center. • Prevents the substrate from entering the active site. • Prevents the catalytic activity. • Examples: insecticides and nerve gases inhibit the enzyme acetylcholinesterase (needed for nerve conduction).
Irreversible inhibitor DFP (diisopropyl fluorophosphate) • DFP forms a covalent bond with the OH group of the amino acid serine in the active site of the enzyme acetylcholinesterase. • Acetylcholinesterase is inhibited. • The transmission of nerve impulses is blocked. • Paralysis occurs.
Irreversible Inhibition • In irreversible inhibition, a substance destroys enzyme activity by bonding with R groups at the active site.
Zymogens Zymogens (proenzymes): • Are inactive forms of enzymes. • Are activated when one or more peptides are removed. • Such as proinsulin is converted to insulin by removing a small peptide chain.
Digestive Enzymes Digestive enzymes are: • Produced as zymogens in one organ and transported to another such as the pancreas when needed. • Activated by removing small peptide sections.
Allosteric Enzymes • An allosteric enzyme is an enzyme in a reaction sequence that binds a regulator substance. • A positive regulator enhances the binding of substrate and accelerates the rate of reaction. • A negative regulator prevents the binding of the substrate to the active site and slows down the rate of reaction.
Feedback Control In feedback control: • A product acts as a negative regulator. • An end product binds with the first enzyme (E1) in a sequence, when sufficient product is present.
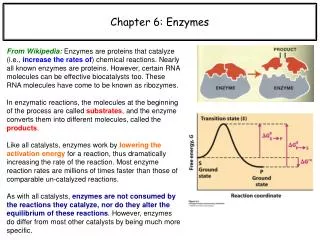
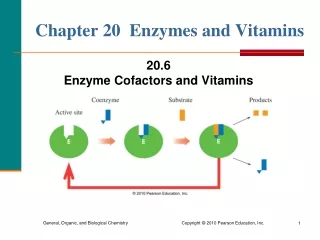
Enzyme Cofactors • A simple enzyme is an active enzyme that consists only of protein. • Many enzymes are active only when they combine with cofactors such as metal ions or small molecules. • A coenzyme is a cofactor that is a small organic molecule such as a vitamin.
Metal Ions as Cofactors • Many active enzymes require a metal ion. • Zn2+, a cofactor for carboxypeptidase, stabilizes the carbonyl oxygen during the hydrolysis of a peptide bond.