Understanding Basic Concepts of Chemistry and Soil Composition
This overview explores the fundamental concepts of chemistry, focusing on the nature of soil, elements, compounds, and reactions. It discusses the components of soil, the structure of glucose, and the identification of pure substances. Additionally, it looks at the properties of elements and compounds, insights into chemical symbols, and the behavior of metals when reacting. Finally, it examines how environmental conditions impact physical properties, such as weight in space exploration. Perfect for students and those looking to refresh their knowledge in chemistry.

Understanding Basic Concepts of Chemistry and Soil Composition
E N D
Presentation Transcript
1. Soil is made up of pieces of rocks, wood, decayed plants, and many other materials. Soil is an example of an atom. an element. a compound. a mixture.
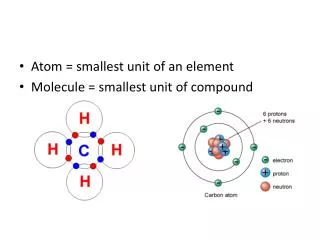
2. The chemical formula for glucose is C6H12O6. How many different elements make up one molecule of glucose? • 1 • 3 • 12 • 24
3. A group of identical atoms would be classified as • a solution. • a compound. • an element. • a mixture.
4. In order to react with other elements, Group 1 metals usually • accept one electron. • give up one electron. • give up one proton. • accept one proton.
5. What property of a space probe will change if a probe from Earth lands on Jupiter? • Mass • Color • Luster • Weight
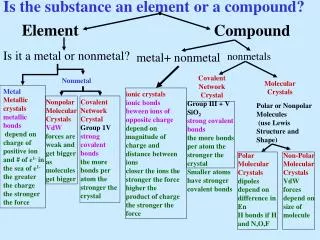
6. How do compounds differ from elements? • Compounds contain only one kind of atom and elements contain more than one kind of atom. • Elements contain only one kind of atom and compounds contain more than one kind of atom. • Compounds form only ionic bonds. • Compounds form only covalent bonds.
7. Which statement is MOST accurate? • A pure substance contains just one kind of matter. • A pure substance has the same composition throughout. • A pure substance can be broken down by physical means. • A pure substance is less stable than a compound.
8. A student observes an element that is solid, shiny, and can be bent into a new shape. The element is probably • Carbon. • Oxygen. • Hydrogen. • Aluminum.
9. Which of the following is a chemical symbol representing an element? • C • CO • CO2 • H2O
10. Radium is an element found in Group 2 and Period 7. In a normal radium atom, how many electron energy levels are present? • 2 • 6 • 7 • 8