Avogadro’s Law
Avogadro’s Law. What is Avogadro’s Law. Avogadro’s Principle – equal volumes of gases at the same temperature and pressure contain equal numbers of particles. Avogadro’s Formula. Formula : V 1 = V 2 n 1 n 2 This is a direct relationship!

Avogadro’s Law
E N D
Presentation Transcript
What is Avogadro’s Law • Avogadro’s Principle – equal volumesof gases at the same temperature and pressure contain equal numbers of particles
Avogadro’s Formula Formula: V1= V2n1n2 • This is a direct relationship! • So if the amount of gas increases, then the volume will ___________. If the amount of gas decreases, then the volume will __________. * n represents the amount of gas increase decrease
Graph for Avogadro’s Law What Laws have we learned were also direct relationships, in which their graphs were similar to Avogadro’s?
Avogadro’s Law • Molar Volume – for a gas, this is the volume that one mole of that gas occupies at STP (STP is standard temperature and pressure which is 0oC and 1 atm) • Avogadro showed experimentally that 1 mole of any gas will occupy a volume of 22.4L at STP **Conversion Factor: 1 mol (any gas) = 22.4 L at STP** (This is on your formula chart under constants and conversions!)
Avogadro’s Law: Example 1 Calculate the volume that 0.881 moles of oxygen gas at STP will occupy. (This can be solved using the Avogadro’s law formula or using the dimensional analysis method)
Avogadro’s Law: Example 1 Answer • Formula: V1= V2 n1 n2 • What do we know? V1 = ? L n1 = 0.881 mol O2 V2 = 22.4 L n2 = 1 mol O2 Remember,at STP: 22.4 L/mol Second way to solve, using the dimensional analysis: .881 mol O2 | 22.4 L = | 1 mol O2 First way to solve, using the formula: V1 = 22.4 L .881 mol O2 1 mol O2 After cross multiplying you end up with V1(1 mol O2) = (22.4 L)(.881 mol O2) 1 mol O2 1 mol O2 V1 = ___________ --------- 19.73 L --------- ---------- ---------- ----------- ---------- 19.73 L
Avogadro’s Law: Example 2 How many grams of N2 will be contained in a 2.0 L flask at STP?
Avogadro’s Law: Example 2 Answer • Formula: V1 = V2 n1 n2 • What do we know? V1 = 2.0 L n1 = ? g N2 V2 = 22.4 L n2 = 1 mol N2 Remember,at STP: 22.4 L/mol First , solve for the number of moles of N2: 2 L N2| 1 mol N2 = .089 mol N2 | 22.4 L N2 Then, use dimensional analysis to convert from moles of N2 to grams of N2: .089 mol N2| 28.014 g N2 = 2.50 g N2 | 1 mol N2
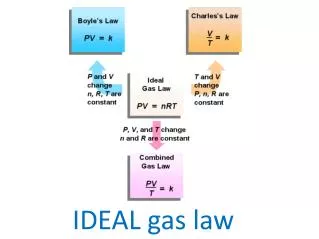
What is the Ideal Gas Law? It is a good approximation to the behavior of many gases under many conditions
Ideal Gas Law Formula • Formula: PV = nRT (called Piv-Nert formula) • R is called the Ideal Gas Constant • R is dependent on the units of the variables for P, V, and T • Temperature is always in Kelvin • Volume is always in Liters • Pressure is either in atm, mmHg, or kPa. Because of the different pressure units, there are 3 possibilities of the ideal gas constant (refer to the EOC Chart under constants and conversions) • Example: R = 0.0821 (Liters)(atm) (moles)(Kelvin) *We would use this value for R if the given pressure’s units are in atm*
Ideal Gas Law Simulation http://phet.colorado.edu/en/simulation/gas-properties
Ideal Gas Law Example 1(using moles) If the pressure by a gas at 30oC in a volume of .05 L is 3.52 atm, how many moles of the gas is present? **To know which R value to use, look at what units pressure is in**
Ideal Gas Law Example 1 Ans(finding moles) • Formula: PV = nRT • What do we know? P = 3.52 atm V = .05 L n = ? moles R = T = 30oC + 273 = 303K What R value will we use? (Hint: Look at the units for pressure) 0.0821 (L*atm)/(mol*K) Now lets plug in the information and solve: P * V = n * R * T (3.52)(.05) = n(.0821)(303) (.0821)(303) (.0821)(303) n = ______________ -------- ------- -------- ----- .0071 mol
Ideal Gas Law Example 2(finding grams) Avogadro’s Law allows us to write a gas law that is valid not only for any P, V, or T but also for any mass of any gas! Example: Calculate the grams of N2gas present in a 0.600 L sample kept at 1.00 atm and a temperature of 22.0oC.
Ideal Gas Law Example 2 Answer(using moles) • Formula: PV = nRT • What do we know? P = 1.00 atm V = 0.600 L n = ? g N2 R = T = 22.0oC + 273 = 295 K What R value will we use? (Hint: Look at the units for pressure) Now lets plug in the information and solve: P * V = n * R * T (1.00)(.600) = n(.0821)(295) (.0821)(295) (.0821)(295) n = ______________ 0.0821 (L*atm)/(mol*K) ------- ------- -------- ----- .025 mol N2 But, they want the answer in grams, so we need to do dimensional analysis using molar mass: .025 mol N2 | 28.014 g N2 = __________ | 1 mol N2 --------- .694 g N2 ----------
Ideal vs. Real Gas • An ideal gas obeys all the assumptions of the kinetic molecular theory. (Atoms or molecules are non-interacting particles, etc.) • NO gas is IDEAL (ideal gas doesn’t exist). They all take up space and interact with other molecules (attraction, repultion) but most gases will behave like ideal gases at the right conditions of temp. and pressure. • Real gases do NOT behave like ideal gases at extremely high pressures and extremely low temperatures.
Explanation • Ideal gases do not have molecular volume and show no attraction between molecules at any distance • Real gas molecules have volume and show attraction at short distances. High P will bring the molecules very close together. This causes more collisions and also allows the weak attractive forces to come into play. With low temperatures (close it gas’s liquefication point), the molecules do not have enough energy to continue on their path to avoid the attraction.
Dalton’s Law Analogy In other words, Dalton’s Law of Partial Pressure would look like this:
Dalton’s Law Formula Formula: Pt= P1 + P2 + P3 + ... Pt = the total pressure P1= Partial Pressure of gas ‘1”, P2 = Partial Pressure of gas “2,” etc. **ALLUnits,MUSTbe the same for each pressure!!!**
What is Dalton’s Law? • Dalton’s Law of Partial Pressures – the total pressure exerted on a container by several different gases is equal to the sum of the pressures exerted on the container by each gas • (Partial pressure of a gas in a mixture of gases is the pressure which that gas would exert if it were the only gas present in the container) • Dalton’s Law of Partial Pressure assumes each gas in the mixture is behaving like an ideal gas
Demo Video’s Simulation (or demo - or show both): Collecting a Gas Over Water http://www.kentchemistry.com/links/GasLaws/dalton.htm Demo - Gas collection over water (or use Butane gas from a cigarette lighter) http://www.youtube.com/watch?v=UQRCUxrLU1c&edufilter=QMQXe2SzEucYVMXVK3i8VQ A more exciting Demo – Oxygen Collection Over water http://www.youtube.com/watch?v=2Tj3Ir0brco&edufilter=QMQXe2SzEucYVMXVK3i8VQ
Volume Ratios All the gas laws you learned so far can be applied to calculate the stoichiometryof reactions in which gases are reactants or products.
For Example 2 C4H10(g) + 13 O2(g) 8 CO2(g) + 10 H2O(g)
We already know that the coefficients in a balanced chemical equation represent the # of moles of the reactants and products in the reaction: ___2___moles of C4H10reacts with __13__moles of O2 to produce ___8___ moles of CO2 and __10__moles of H2O
Remember Avogadro’s principle states that equal volumes of gases at the same temperature and pressure contain equal numbers (or moles) of particles. **1 mole of any gas occupies a volume of 22.4L at STP**
So, the coefficients in a balanced equation also show the relationships among the volumes of any gaseous reactants or products. Ex: ___2___ L of C4H10 react with ___13___L of O2 to produce____8____L of CO2 and ____10____L of H2O
Calculations InvolvingOnly Volume (the molar ratios are also volume ratios for gases) What volume of oxygen gas is needed to complete the combustion of 4.0L of propane gas (C3H8) at STP? _____C3H8(g) + _____O2(g) _____CO2(g) + _____H2O(g) 1 5 3 4
__1___C3H8(g) + __5___O2(g)__3___CO2(g) + __4___H2O(g) 4 L ? L Start with what you are given, then use Dimensional Analysis to get the answer: 4 L C3H8 | 5 L O2 = | 1 L C3H8 --------- 20 L O2 ---------
You Try! What volume of oxygen is needed to react with solid sulfur to form 3.5 L SO2? Assume constant pressure and temperature. ___O2+ ___S ___SO2
Calculations InvolvingMass and Volume (use the PT when converting grams to moles) If 5.0 L of nitrogen gas reacts completely with hydrogen gas at a temperature of 298K and a pressure of 3.0 atm, how many grams of ammonia (NH3) are produced? _____N2(g) + _____H2(g) _____NH3(g) 1 3 2
You Try! Ammonia nitrate is a common ingredient in chemical fertilizers. Use the reaction shown to calculate the mass of solid ammonium nitrate that must be used to obtain 0.100 L of dinitrogen oxide gas at STP. ___NH4NO3(s) ___N2O(g) + ___H2O(g)