G (molar mass)
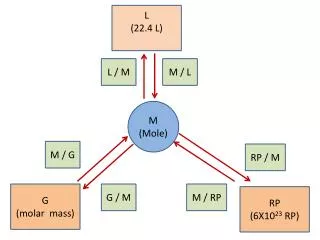
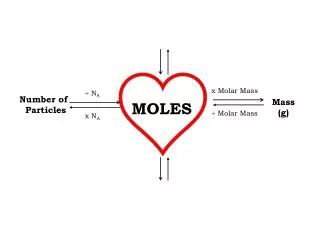
L (22.4 L). L / M. M / L. M (Mole). M / G. RP / M. G (molar mass). G / M. M / RP. RP (6X10 23 RP). Things you must know:. 1 mole = the molar mass of a substance (G) 1 mole = 6.02 X 10 23 representative particles (RP) (atoms, molecules, FU)

G (molar mass)
E N D
Presentation Transcript
L (22.4 L) L / M M / L M (Mole) M / G RP / M G (molar mass) G / M M / RP RP (6X1023 RP)
Things you must know: 1 mole =the molar mass of a substance (G) 1 mole =6.02 X 1023 representative particles (RP) (atoms, molecules, FU) 1 mole =22.4 liters of a gas (L)
Other things you must know • Polyatomic ions • Group B ions (cuprous, ferric, etc.) • Writing formulas from names • Balancing ionic formula charges • Scientific notation • Representative particles (atoms, molecules, FU) • Read chapter 6 and 7
How to start a problem • Write the balanced formulas • Calculate the molar mass • Determine the given – put it on the left • Determine the unit of what you are looking for – put it on the right • Fill in the center with conversion factors
How many atoms are in 5.12 moles of magnesium phosphate? (scientific notation)
How many liters in 238 g CO2? 238 G 1 1 M 44 G 22.4 L 1 M 121 L
How many grams in 1.806 X1025 formula units of aluminum oxide ?
How many grams in 1.806 X1025 formula units of aluminum oxide ? 1.806 X1025 FU 1 1 M 6.02X1023 FU 102 G 1 M 3060 G
How many molecules in .0684 l nitrogen gas? .0684 L 1 1 M 22.4 L 6X1023 FU 1 M 1.8 X 1021 FU