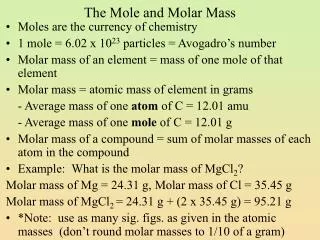Molar Mass
Molar Mass. Molecular Mass. The molecular mass of a substance is the mass in atomic mass units ( amu ) of all the atoms in a given molecule. It is more commonly referred to as molecular weight and abbreviated as MW.

Molar Mass
E N D
Presentation Transcript
Molecular Mass • The molecular mass of a substance is the mass in atomic mass units (amu) of all the atoms in a given molecule. • It is more commonly referred to as molecular weight and abbreviated as MW. • An atomic mass unit is defined as 1/12 the mass of the carbon-12 isotope. The old symbol was amu, while the symbol "u" is now used. Carbon-12 is defined as weighing exactly 12 u per atom.
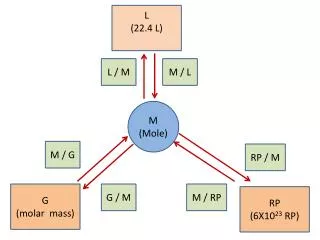
Molar Mass • Counting atoms or molecules is very difficult since they are so small. But it is possible to determine the mass of large amounts of them using a balance. • The mass of one mole of a substance is called its molar mass with units of g/mol. • The molar mass of a substance can be found by dividing the mass in grams by the number of moles of the substance, thus giving us the mass per mole of the substance.
Calculating Molar Mass Four steps to calculating a substance's molar mass: • Determine how many atoms of each different element are in the formula. • Look up the atomic mass of each element in a periodic table. • Multiply step one times step two for each element. • Add the results of step three together.
Example Tip: It is best to carry all of the available decimal places, and round only the final answer to the appropriate decimal place. Calculate the molar mass of Al(NO3)3. • There is 1 atom of AlThere are 3 atoms of NThere are 9 atoms of O • The atomic mass of Al is 26.9815 g/molThe atomic mass of N is 14.0067 g/molThe atomic mass of O is 15.9994 g/mol • For Al, 1 mol x 26.9815 g/mol = 26.9815 g/molFor N, 3 moles x 14.0067 g/mol = 42.0201 g/molFor O, 9 moles x 15.9994 g/mol = 143.9946 g/mol • For the entire molecule:26.9815 + 42.0201 + 143.9946 = 212.9962 g/mol The molar mass of aluminum nitrate is 212.996 g/molNote: The decimal places of the answer are consistent with the decimal places in the atomic masses, your periodic table might be less precise (less decimals).
Check Your Understanding • Which of the following chemical formulas has a molar mass of 34.0146 g/mol?a) H2Ob) H2O2c) HO2d) CH2O • What is the molar mass of aluminum sulphate?
Mole Conversions: Converting moles to grams Three steps to converting moles of a substance to grams: • Determine how many moles are given in the problem. • Calculate the molar mass of the substance. • Multiply step one by step two.
Example 2 Calculate how many grams are in 0.7 mol of H2O2 (hydrogen peroxide which is used to sterilize wounds). Step one: The problem will tell you how many moles are present. Look for the word "mole" or the unit "mol." The number immediately preceding it will be how many moles. In this question, 0.7 mol are given Step two: You need to know the molar mass of the substance; hydrogen peroxide contains 2 H and 2 O atoms in each molecule. The molar mass of H2O2 is 34.0146 g/mol Step three: You multiply the mol given by the substance's molar mass:0.7 mole x 34.0146 g/mol = 23.8 g So, there are 23.8 g in 0.7 mol of H2O2
Example 3 Convert 2.5 mol of KClO3 to grams. 2.50 mol is given in the problem.MM (KClO3) = 122.55 g/mol (the combined mass of 1 K, 1 Cl and 3 O atoms) 2.5 mol x 122.55 g/mol = 306.375 g There are 306.38 g in 2.5 mol of KClO3
Check Your Understanding • If you had 1.5 mol of each of the following compounds, which would have a mass of 135.8 g?a) H2O2b) H2SO4c) KClOd) MgCl2 • What is the mass of 3.40 x 10-2 mol of Na2CO3?
Mole Conversions: Converting Grams to Moles Three steps to converting moles of a substance to grams: • Determine how many grams are given in the problem. • Calculate the molar mass of the substance. • Divide step one by step two.
Example 4 Convert 25.0 g of KMnO4 to moles. Step one: The question will tell you how many grams are present. Look for the unit of grams or g. The problem gives you 25.0 g. Step two: You need to know the molar mass of the substance. The molar mass of KMnO4 is 158.034 g/mol. Step three: You divide the grams given by the substance's molar mass:The answer is 25.0 g / 158.034 g/mol = 0.158 mol. 25.0 g of KMnO4 represents 0.158 mol.
Example 5 Calculate how many moles are in 17.0 g of H2O2. 17.0 grams are given in the question. MM (H2O2) = 34.0146 g/mol. Therefore, 17.0 g of H2O2 corresponds to 0.5 mol.
Check Your Understanding • Which of the following represents 0.219 mol?a) 2.00 g of H2Ob) 75.57 g of KBrc) 100 g of KClO4d) 8.76 g of NaOH • How many moles are present in 57.68 g of calcium nitrite?
Mole Conversions: Calculating the Mass of One Molecule To calculating the mass of one molecule: • Calculate the molar mass of the substance. • Divide it by Avogadro's number.
Example 6 What is the mass of one molecule of H2O? Step one: The molar mass of water is 18.015 g/mol. Step two: Divide the substance's molar mass by Avogadro's number. So, one molecule of H2O has a mass of 2.99x10-23 g
Example 7 Calculate the mass (in grams) of one molecule of acetic acid, CH3COOH. MM (CH3COOH) = 60.06 g/mol So, one molecule of acetic acid has a mass of 9.98 x 10-23 g.
Check Your Understanding • What is the mass (in grams) for one molecule of UF6?