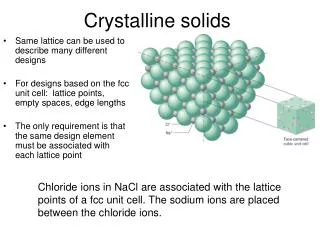
Crystalline Solids
DESCRIPTION
Crystalline Solids. p. 459. Macroscopic structure depends upon microscopic structure. Pyrite (FeS 2 , fool’s gold). Fluorite (CaF 2 ). Amethyst (SiO 2 ) – quartz + Fe & Mn. Crystal terms. Crystal – Lattice – Unit cell – Edge length (d o ) – . face. edge. corner.
1 / 29
Download Presentation


Télécharger la présentation 

Crystalline Solids
An Image/Link below is provided (as is) to download presentation
Download Policy: Content on the Website is provided to you AS IS for your information and personal use and may not be sold / licensed / shared on other websites without getting consent from its author.
Content is provided to you AS IS for your information and personal use only.
Download presentation by click this link.
While downloading, if for some reason you are not able to download a presentation, the publisher may have deleted the file from their server.
During download, if you can't get a presentation, the file might be deleted by the publisher.
E N D
Presentation Transcript
Pyrite (FeS2, fool’s gold) Fluorite (CaF2) Amethyst (SiO2) – quartz + Fe & Mn
Crystal terms • Crystal – • Lattice – • Unit cell – • Edge length (do) – face edge corner
Packing in Crystals • Atoms and ions are represented by spheres. • The spheres stack in different patterns three-dimensionally. • The close packing of spheres maximizes intermolecular force. • The patterns of stacking result in different types of unit cells and lattices.
Alloys Intermetallic Compounds
More Related

Audio
Live Player