Transcription…
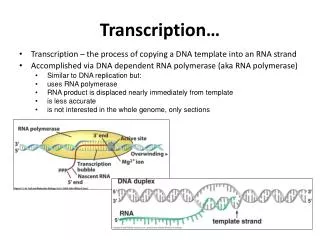
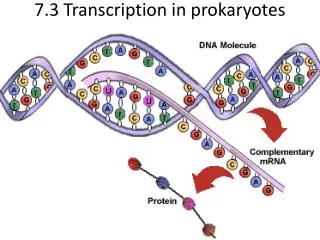
Transcription…. Transcription – the process of copying a DNA template into an RNA strand Accomplished via DNA dependent RNA polymerase (aka RNA polymerase) Similar to DNA replication but: uses RNA polymerase RNA product is displaced nearly immediately from template is less accurate

Transcription…
E N D
Presentation Transcript
Transcription… • Transcription – the process of copying a DNA template into an RNA strand • Accomplished via DNA dependent RNA polymerase (aka RNA polymerase) • Similar to DNA replication but: • uses RNA polymerase • RNA product is displaced nearly immediately from template • is less accurate • is not interested in the whole genome, only sections
Transcription… • By the end of this series, you should be able to explain much of this animation • http://www.as.wvu.edu/~dray/219files/Transcription_588x392.swf
Bacterial RNA polymerase 50 - 100 nucleotides/sec • Most genes are transcribed simultaneously by numerous polymerases • Polymerase moves along DNA in 3' —> 5' direction • Complementary RNA constructed in ____ direction • RNAn + NPPP —> RNAn+1 + PPi
Transcription • How does the polymerase know where to start? • Promoter = the assembly point for the transcription complex • RNA polymerases cannot recognize promoters on their own - transcription factors • Transcription factors - enzymes have evolved to recognize (physically interact with) specific DNA sequences and with other proteins
Prokaryotic Gene Regulation • Preliminaries • DNA binding proteins • Minor vs. Major grooves • Bases are exposed in the grooves, providing binding sites • Each bp provides characteristic binding sites A – H acceptor, D – H donor, H – nonpolar H, M – methyl group
Prokaryotic Gene Regulation • Protein-nucleic acid binding • Most proteins to be discussed bind specific DNA sequences • Most commonly via α-helix insertion into major groove(s) • H donor/acceptors • + charge on helix interaction with phosphate backbone • Helix-turn-helix motif • 1st DNA binding domain identified
Transcription • Three-phase progression • Initiation – • Promoter recognition and binding • DNA melting • Transcript initiation • Elongation • mRNA produced • Termination • Polymerase and RNA released
Transcription • Bacterial transcription • Promoters • Core enzyme (α2ββ’ω) will begin transcription just about anywhere • σ provides specificity • Core + σ = holoenzyme • E. coli σ70 • σ70 promoters • 2 conserved sequences of 6 bp each • -10, -35
Transcription • Bacterial transcription • Promoters • Not all -10 and -35 sequences are identical • Consensus sequences • -35 – TTGACA • -10 - TATAAT
Transcription • Prokaryotic Transcription • Bacterial promoters are located just upstream of the RNA synthesis initiation site • The nucleotide at which transcription is initiated is called +1; the preceding nucleotide is –1 • DNA preceding initiation site (toward template 3' end) are said to be upstream • DNA succeeding initiation site (toward template 5' end) are said to be downstream
Transcription • Prokaryotic Transcription • One RNA polymerase with 5 subunits tightly associated to form core enzyme • Core enzyme minus sigma (σ) factor will bind to any DNA. • By adding σ, RNA pol will bind specifically to promoters (-10 & -35 sequences) • Transcription begins de novo • Requires stable interaction b/t base and template while second base is recruited
Transcription • Bacterial transcription • Three models of initial transcription • How to explain? • Transient excursion – Pol moves along DNA chain • Inchworming – • Scrunching – DNA pulled in • Experiments demonstrate: • Pol remains stationary on promoter • Pol subunits remain stationary relative to one another • Suggests that ‘scrunching’ is it • As DNA/RNA ‘piles up’ inside the polymerase, it creates pressure which contributes to forcing the polymerase off of the promoter
Transcription • Bacterial transcription • Several false starts before elongation begins • ≤ 9bp • ‘Escape’ - transcription of 10+ bp leading to elongation phase • 10+ bp too long to remain hybridized to tempate • Pol must break interactions with promoter and regulatory factors • σ ¾ linker must be ejected from RNA exit channel • ‘Scrunched’ DNA likely provides the energy to break pol-promoter interactions and dislodge σ
Transcription • Bacterial transcription • Termination • Rho (ρ) dependent • ATPase activity leads to translocation along transcript
Transcription • Bacterial transcription • Termination • ρ-dependent • Rut (ρ utilization) sites – not well-defined but sequence dependent • http://archive.microbelibrary.org/microbelibrary/files/ccImages/Articleimages/sandrin/rhodependent.htm • http://highered.mcgraw-hill.com/sites/dl/free/0072835125/126997/animation21.html
Transcription • Bacterial transcription • Termination • ρ-independent • Two sequence elements • Inverted repeat • ~8 A:T stretch • Hairpin formation disrupts polymerase function • A:U binding weak
Transcription • Eukaryotic vs. Prokaryotic Transcription • Much of what we know is derived from studies of RNA pol II from yeast • 1. Seven more subunits than its bacterial RNA pol • 2. The core structure & the basic mechanism of transcription are virtually identical • 3. Additional subunits of eukaryotic polymerases are thought to play roles in the interaction with other proteins • 4. Eukaryotes require a large variety of accessory proteins or transcription factors (TFs)
Transcription • Eukaryotic transcription • Contrasts with prokaryotes • Three polymerases • RNA polymerase I (RNA pol I) - synthesizes the larger rRNAs (28S, 18S, 5.8S) • RNA polymerase II (RNA pol II)- synthesizes mRNAs & most small nuclear RNAs (snRNAs & snoRNAs) • RNA polymerase III (RNA pol III) - synthesizes various small RNAs (tRNAs, 5S rRNA & U6 snRNA) • Multiple general transcription factors (GTFs) • Additional regulatory elements (enhancers, chromatin modifiers, silencers, insulators, etc.) • RNA pol II – transcribes mRNA
Transcription • Eukaryotic transcription • RNA pol II core promoters • Four different sequence elements • TFIIB recognition element (BRE box) • TATA box • Initiator box (Inr) • Downstream promoter elements (DCE, DPE, MTE) • Various combinations in most Pol II promoters • All not necessary • Inr most common
Transcription • Eukaryotic transcription • Preinitiation complex formation • TBP component of TFIID • β sheet inserted into minor groove, bending DNA 80° • Specificity via phenylalanine chain intercalation on flanks of sequence
Transcription • Eukaryotic transcription • Preinitiation complex formation • TFIID TFIIA TFIIB TFIIF TFIIE TFIIH
Transcription • Eukaryotic transcription • Preinitiation complex formation • TFIID – initial binding and recruitment • TFIIA – clamp • TFIIB - recruitment of pol II, may insert into RNA exit channel • TFIIF – stabilize complex, required for recruitment of TFIIE/H • TFIIE – recruit and regulate TFIIH • TFIIH – kinase • Largest GTF • Phosphorylates carboxy-terminal domain (CTD) of pol II • http://www.crocoduck.bch.msstate.edu/BCH4713/Transcription.wmv
RNA processing – mRNA • 5’ cap • The raw transcript will be immediately degraded in the cytoplasm so it must be marked and protected • Possible/known functions of 5’ cap • May prevent exonuclease digestion of mRNA 5' end, • Aids in transport of mRNA out of nucleus • Important role in initiation of mRNA translation
RNA processing – mRNA • mRNA processing – Splicing • Requires break at 5' & 3' intron ends (splice sites) & covalent joining of adjacent exons (ligation) • http://www.as.wvu.edu/~dray/219files/mRNASplicingAdvanced.wmv • Why introns? • Disadvantages – extra DNA, extra energy needed for processing, extra energy needed for replication • Advantages – modular design allows for greater variation and relatively easy introduction of that variation
RNA processing – mRNA • mRNA processing – Splicing • Splicing MUST be absolutely precise • Most common conserved sequence at eukaryotic exon-intron borders in mammalian pre-mRNA is G/GU at 5' intron end (5' splice site) & AG/G at 3' end (3' splice site)
RNA processing – mRNA • mRNA processing – Splicing • Sequences adjacent to introns contain preferred nucleotides that play an important role in splice site recognition
RNA processing – mRNA • mRNA processing – Splicing • Nuclear pre-mRNA (common) • snRNAs + associated proteins = snRNPs • snRNAs – 100-300 bp • U1, U2, U4, U5, U6 • 3 functions for snRNPs • Recognize sites (splice site and branch point site) • Bring these sites together • Catalyze cleavage reactions • Splicosome – the set of 5 snRNPs and other associated proteins • Summary movie available at: • http://www.as.wvu.edu/~dray/219files/mRNAsplicing.swf
RNA processing – mRNA • mRNA processing – Splicing • 1. U1 and U2 snRNPs bind via complementary RNA sequences • Note the A bulge produced by U2 • U2 is recruited by proteins associated with an exon splice enhancer (ESE) within the exon
RNA processing – mRNA • mRNA processing – Splicing • 2. U2 recruits U4/U5/U6 trimer • U6 replaces U1, U1 and U4 released • U5 binds to upstream exon
RNA processing – mRNA • mRNA processing – Splicing • 3. U6 catalyzes two important reactions • Cleavage of upstream exon from intron (bound to U5) • Lariat formation with A bulge on intron • Exons are ligated • U2/U5/U6 remain with intron
DNA/RNA Structure OH OH NB NB NB NB DNA RNA OH O OH OH P O C OH O P P P O O O C C C O OH OH
RNA processing – mRNA • mRNA processing – Splicing • http://www.crocoduck.bch.msstate.edu/BCH4713/ch13_group_II_introns.html • Several lines of evidence suggest that it is the RNA in the snRNP that actually catalyzes the splicing reactions • 1. Pre-mRNAs are spliced by the same pair of chemical reactions that occur as group II (self-splicing) introns • 2. The snRNAs needed for splicing pre-mRNAs closely resemble parts of the group II introns • Proteins likely serve supplemental functions • 1. Maintaining the proper 3D structure of the snRNA • 2. Driving changes in snRNA conformation • 3. Transporting spliced mRNAs to the nuclear envelope • 4. Selecting the splice sites to be used during the processing of a particular pre-mRNA
Transcription • Polyadenlyation • The poly(A) tail – 3' end of most mRNAs contain a string of adenosine residues (100-250) that forms a tail • Protects the mRNA from degradation • AAUAAA signal ~20 nt upstream from poly(A) addition site • Poly(A) polymerase, poly(A) binding proteins, and cleavage factors are involved • Simple animation on website
Transcription • Eukaryotic transcription • Termination • Torpedo model • Post-cleavage RNA is uncapped • Recognized by Rnase (Xrn2 in humans) • Again, CTD bound • Highly processive, displaces Pol II
Transcription • Eukaryotic RNA pol I • rRNA transcription • SL1 = TBP + three TAFs
Transcription • Eukaryotic RNA pol III • tRNA and 5S transcription • Internal promoter sequences
Prokaryotic Gene Regulation • Principles • Activators and repressors • DNA binding proteins • Primary level of action – transcription initiation • Activators enhance RNA polymerase binding • Repressors block RNA polymerase binding • Many regulatory proteins work via allostery
Prokaryotic Gene Regulation • Principles • Action at a distance • Interaction b/t distantly binding proteins accommodated by DNA loops • Often aided by architectural proteins
Prokaryotic Gene Regulation • Control of Gene Expression: Prokaryotes • The operon - in bacteria, genes for enzymes of metabolic pathway are usually clustered in functional complex under coordinate control • Terminology: • 1. Genes - code for operon enzymes; usually adjacent to each other; turn on one, turn on all • 2. Promoter • 3. Operator – typically resides adjacent to or overlapping with the promoter; repressor protein binding site • 4. Repressor - gene regulatory protein; binds with high affinity to operator • 5. Regulatory gene - encodes repressor protein
Prokaryotic Gene Regulation • The operon… • The repressor is key to operon expression; if it binds to operator; it shields promoter from polymerase & prevents transcription • 1. Repressor binding to operator depends on conformation, which is regulated by a key compound in the metabolic pathway (lactose or tryptophan) • 2. Concentration of key metabolite determines if operon is active or inactive at any given time
Prokaryotic Gene Regulation • The lac operon… • An inducible operon – the presence of a key substance induces the transcription of the genes. • Regulates production of the enzymes needed to degrade lactose in bacterial cells • Genes in the lac operon • 1. z gene - encodes β-galactosidase • 2. y gene - encodes galactoside permease; promotes lactose entry into cell • 3. a gene - encodes thiogalactoside acetyltransferase; its physiological role is unclear
Prokaryotic Gene Regulation • Prokaryotic gene expression… • Lactose (disaccharide) - made of glucose & galactose • Oxidation provides the cell with metabolic intermediates & energy • The β-galactoside linkage is broken in the first step of catabolism - β-galactosidase
Prokaryotic Gene Regulation • Control of Gene Expression: Prokaryotes • Prokaryotes live in constantly changing environment • It is advantageous for cells to use available resources in most efficient way so regulate responses • Thus, they respond by selective gene expression • If lactose is absent—> β-galactosidase not needed & not present (<5 copies of enzyme, 1 of the corresponding mRNA) • If lactose is present —> enzyme levels rise ~1000-fold in a few minutes; lactose has induced the synthesis of β-galactosidase
Prokaryotic Gene Regulation • The lac operon… • 1. If lactose is present in medium, it enters cell, binds lac repressor, changing its shape. Lactose acts as an inducer • 2. Lactose-bound repressor cannot bind operator DNA • 3. If lactose levels fall, it dissociates from repressor, changing repressor back to active shape • 4. Repressor binds operator and physically blocks polymerase from reaching structural genes, turns off transcription • Lac operon movie
Prokaryotic Gene Regulation • The lacoperon • Lac repressor binding
Prokaryotic Gene Regulation • The lacoperon • Both bind using similar amino acid motifs • Helix-turn-helix • Recognition helix interacts with DNA • λ-repressor example
Transcription... • Control of Gene Expression: Prokaryotes • Tryptophan - essential amino acid needed for protein synthesis; if it is not in the growth medium, it must be produced by bacterium • 1. In its absence, cells contain enzymes & their mRNAs needed to make tryptophan • 2. If tryptophan is available in medium, bacteria don't need enzymes to make it; the genes of those enzymes are repressed within a few minutes & the production of the enzymes stops
Transcription... • The trp operon… • A repressible operon – the presence of a key substance represses the transcription of genes. • Repressor is active only if bound to specific factor which functions as a co-repressor (like tryptophan)
Transcription... • The trp operon… • 1. Without tryptophan, operator site is open to binding by RNA polymerase • 2. Production of enzymes that synthesize tryptophan • 3. When tryptophan is available, enzymes of tryptophan synthetic pathway are no longer needed • 4. Increased tryptophan concentration leads to formation of tryptophan-repressor (active repressor) • 5. Repressor binds DNA at operator, blocks transcription • http://bcs.whfreeman.com/thelifewire/content/chp13/1302002.html
The Cell Nucleus… • Control of Gene Expression: Eukaryotes • A single human cell contains enough DNA (6 billion bp) to encode several million different polypeptides • 1. Most of this DNA does not actually code for proteins, mammalian genomes are thought to contain ~30,000 protein-coding genes • 2. A typical mammalian cell may make ~5,000 different polypeptides at any given time • 3. Many of these are made by virtually all cells of the organism • 4. Cells also make proteins unique to its differentiated state; giving the cell its unique characteristics • 5. Regulating eukaryotic gene expression is an extremely complex process, just starting to be understood