STERILITY TEST
STERILITY TEST. Required for all articles or substances to be introduced into raw tissue (injections and ophthalmics ). Sterility testing attempts to reveal the presence or absence of viable micro-organisms in a sample number of containers taken from batch of product.


STERILITY TEST
E N D
Presentation Transcript
STERILITY TEST • Required for all articles or substances to be introduced into raw tissue (injections and ophthalmics). • Sterility testing attempts to reveal the presence or absence of viable micro-organisms in a sample number of containers taken from batch of product. • Based on results obtained from testing the sample a decision is made as to the sterility of the batch. • The test show that samples tested were free from living bacteria and fungi but not viruses.
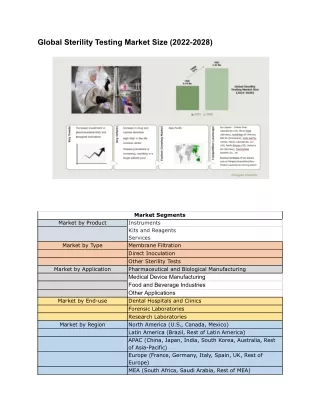
Methods of sterility test • Membrane filtration • Direct inoculation of the culture medium
Direct inoculation of the culture medium • Culture media for sterility test must be: • Capable of initiating and maintaining the vigorous growth of a small number of organisms. • Sterile. • There is no one medium which could detect both bacteria and fungi.
Direct inoculation of the culture medium • The USP recommends two culture media for sterility testing: a) Fluid Thioglycolate Medium (FTM) for detection of both aerobic and anaerobic bacteria. b) Trypticase Soya Broth (TSB) (Fluid soya bean casein digest medium) primarily intended for the culture of both fungi and aerobic bacteria.
Formulation of "FTM" • Yeast extract..........……......... nutrient (growth factor) • Pancreatic digest of casein………nutrient (nitrogen source) • Dextrose……...nutrient (carbon source) and antioxidant • L-cysteine...... antioxidant, support growth of clostridia & neutralize metal preservatives • Sod. thioglycolate……...antioxidant & neutralize metal preservatives. • Agar (0.075%)…… viscosity inducer to reduce oxygen diffusion • Resazurin sod……... redox indicator (pink at high O2 tensioninupper third of container and yellow at low O2 tension in lower 2/3 of the container). • Distilled water pH after sterilization 7.1 0.1
Formulation of "TSB" • Pancreatic digest of casein.................... nutrient • Papaic digest of soya bean ................... nutrient • Dextrose................................................... nutrient • Sod. chloride................................. isotonic agent • Dibasic potassium phosphate....................buffer • Distilled Water pH after sterilization 7.3 0.1
Direct inoculation of the culture medium • Suitable quantity of the preparation to be examined is transferred directly into the appropriate culture medium. • Volume of the product is not more than 10% of the volume of the medium. • Suitable method for aqueous solutions, oily liquids, ointments an creams .
Direct inoculation of the culture medium Procedure: • The container is cleaned with suitable antimicrobial agent and aseptically opened to avoid contamination and hence false positive result. • The specified volume of the sample is withdrawn (1 ml). • Aseptically, one half is added to "FTM" and the other half is added to "TSB". • "FTM" is incubated at 30-35oC (suitable for bacteria). • "TSB" is incubated at 20-25oC (suitable for fungi).
Interpretation of results • If no growth is observed after the incubation period the batch pass the test. • If visible growth is observed the test is repeated. • If again growth is observed the material fails the test. • It is recommended to identify the contaminant and trace it (by staining for bacterial contaminant).
Direct simple stain procedure • Prepare a heat fixed bacterial smear . • Leave to cool. • Cover the film with crystal violet. • Leave for 30 sec. • Wash gently with water. • Dry between 2 filter papers. • Add oil and examine using oil immersion lens.
Staphylococcus aureus • Shape: cocci • Size: small • Arrangement: Clusters, bunches • Color: violet
Escherichia coli • Shape: Rods • Size: Small • Arrangement: Single • Color: violet
Candida albicans • Shape: Oval • Size: Large • Arrangement: Single • Color: violet
Aspergillusniger(wet mount) Head (sporangium) Spores Mycelium (sporangiophore)