Organic Nomenclature
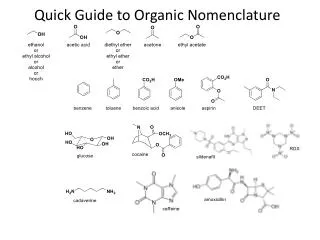
Organic Nomenclature. Compounds in which carbon is the principal element. Writing formulas. . . . Condensed: CH 3 CH(CH 3 )CH = CHCH(CH 3 )CH 3 Kekule’ Skeletal. Hydrocarbons. . . . Contain only hydrogen and carbon. Types:

Organic Nomenclature
E N D
Presentation Transcript
Organic Nomenclature Compounds in which carbon is the principal element.
Writing formulas. . . • Condensed: CH3CH(CH3)CH = CHCH(CH3)CH3 • Kekule’ • Skeletal
Hydrocarbons. . . • Contain only hydrogen and carbon. • Types: • Alkane: single bonds; saturated (largest possible number of hydrogens/carbon atom. [CnH2n+2] • Alkene: one or more carbon-carbon double bond. Unsaturated hydrocarbon. • Alkyne: one carbon-carbon triple bond. Unsaturated hydrocarbon. • Aromatic: carbon atoms connected in a planar ring. Unsaturated hydrocarbon.
Hydrocarbons. . . • Relatively nonpolar. (covalent bonds) • Insoluble in water. (like dissolves like) • Less volatile with increasing molar mass. (because of London dispersion forces)
Alkanes. . . [CnH2n+2] • Methane: CH4 major component of natural gas; home heating; gas stoves; hot water heaters • Ethane: C2H6 • Propane: C3H8 used for home cooking and heating; gas grills • Butane: C4H10 disposable lighters and fuel canisters for gas camping stoves.
Name consists of. . . • 1. Prefix • Where are the substituents? • 2.Parent • How many carbons in the longest chain? • 3. Suffix • Which family? • If two different chains of equal length are present: • choose the one with the larger number of branch points as the parent.
Rules for naming alkanes. . . Longest continuous chain of carbon atoms gives the root name 1. For alkanes, add –ane to the root: Meth: C Eth: C2 Prop: C3 But: C4 2. For alkanes beyond butane, use the Greek root for the number of carbons then add -ane to the end. C-C-C-C-C-C = hexane pent=5; hex=6; hept=7; oct=8; non=9; dec= 10
Substituent Group(s) added in place of a removed hydrogen.
Alkanes as a substituent group. . . Alkyl substituents: 1. Remove a hydrogen from the alkane. C2H6 is ethane 2. Drop the -ane and add -yl. -C2H5 is ethyl
C • C-C-C-C-C-C Positions of substituent groups are specified by numbering the longest chain sequentially. (Use the lowest number for the position of the substituent group) The location and name of the substituent group is followed by root alkane name. 3-methylhexane
Substituents in alphabetical order. Use di-, tri-, etc. to indicate multiple identical substituents.
Practice. . .write skeletal and condensed formulas. • 2,3-dimethylhexane • 2-ethyl-3-methylpentane • 4-ethyl-3,5-dimethylnonane • 4-tert-butylheptane • 2,3-dimethylpentane
Cyclic Alkanes. . . CnH2n • Cycloalkanes • Smaller than five carbons, very reactive. • Rings of carbon atoms. • Isomers • Cis: both substituents are on the same side of the ring. • Trans: substituents are on opposite sides of the ring.
Reactions of Alkanes. . . . • Combustion: react with oxygen produce carbon dioxide and water • Substitution: halogen atoms replace hydrogen atoms • Dehydrogenation reactions:hydrogen removed forms double bond there unsaturated hydrocarbon is the product
Alkenes. . . • 1. Root hydrocarbon (longest chain containing double bond) name ends in –ene. • 2. More than three carbon atoms, the location of the double bond is indicated by the lowest numbered carbon atom in the bond. If equidistant: begin at end closer to substituent group. • CH2=CHCH2CH3 1-butene • CH3CH=CHCH3 2- butene • 3. More than one double bond • Indicate the position of each and use –diene, triene, tetraene. . .
Cycloalkenes. . . • Name same as alkenes. • Number so double bond is between C1 and C2 and the first substituent has the lowest number possible.
Practice. . .write the skeletal formula and name. • CH2=CHCH(CH3)C(CH3)(CH3)CH3 • CH3CH2CH=C(CH3)CH2CH3 • CH3CH=CHCH(CH3)CH=CHCH(CH3)CH3
Practice. . . • 2-methyl-1,5-hexadiene • 3-ethyl-2,2dimethyl-3-heptene • 2,3,3-trimethyl-1,4,6-octatriene • 4-tert-butyl-2-methylheptane • 3,4-diisopropyl-2,5-dimethyl-3-hexene
CH3 CH3 CH(CH3)2 CH3 CH3 Practice. . .
Alkynes. . . • Unsaturated hydrocarbons contain atleast one triple carbon-carbon bond. • -yne replaces –ane. • Begin numbering chain at end closest to triple bond. • More than one bond: -diynes, -triynes. • Double and triple bonds: -enynes • Start numbering from end nearer first bond. (if there is a choice. Usually double bonds get lower numbers than triple.)
Practice. . . • CH3CH(CH3)C CCH(CH3)CH3 • CH CC(CH3)(CH3)CH3 • CH3CH=CHCH=CHC CCH3 • CH3CH2C(CH3)(CH3)C CCH2CH2CH3
Reactions of Alkenes and Alkynes. . . • Addition reactions: double bond broken and new bonds formed to atoms added. • Hydrogenation reactions: addition of hydrogen atoms • Halogenation: addition of a halogen • Elimination:one reactant splits apart to give two products. • Substitutions: two reactants exchange parts to give two new products. • Rearrangement:one reactant undergoes a reorganization of bonds and atoms to give a new product.
Aromatic Hydrocarbons. . .Benzene and its structural relatives • Monosubstituted benzene: named as other hydrocarbons, with benzene as parent name. • If substituent larger than ring (six carbons) : benzene becomes substituent group called phenyl. • Disubstituted benzene: ortho:1,2 meta-1,3 para-1,4 • More than two: number the position of each substituent group.
Hydrocarbon derivatives. . . • Alcohols: presence of an –OH (hydroxyl group) • Nomenclature: replace final -e of parent hydrocarbon with –ol. • (parent: longest chain containing –OH) • Position of –OH specified by number. • Number is lowest of substituent groups. • Classified by: number of hydrocarbon fragmennts bonded to carbon where –OH group is attached. • R,R’,R”represent the hydrocarbon fragments.
Alcohols. . . . • Polar • Higher boiling points than expected from molecular weights. • Intermolecular attractions. –OH extensive hydrogen bonding.
Practice. . . • CH3C(OH)(CH3)CH2CH2CH3 • 2methyl-2-pentanol • CH3CH(OH)CH2CH(OH)CH(CH3)2 • 5-methyl-2,4-hexanediol
Practice. . . • 2-ethyl-2-buten-1-ol • 3-cyclohexen-1-ol • trans-3-chlorocycloheptanol • 1,4-pentanediol
CH3 OH CH3 CH2CH2C(OH)(CH3)2 Practice. . . 4,4-dimethylcyclohexanol 2-methyl-4-phenyl-2-butanol
Aldehydes. . . • Nomenclature: remove –e replace with –al. • Longest chain as base name,must contain the carbonyl group. Aldehyde carbon assigned number 1, when subtituent positions are listed in the name. • Bonded to at least one hydrogen atom. • Number indicates position of carbonyl group. • -CHO group attached to a ring, suffix -carbaldehyde.
Practice. . . • CH3CHO • Ethanal • CH3CH2CHO • Propanal • CH3CH(CH3)CH2CH(CH2CH3)CHO • 2-ethyl-4-methylpentanal
CHO CHO Practice. . . Cyclohexanecarbaldehyde benzenecarbaldehyde
Ketones. . . • Contain carbonyl group bonded to two carbon atoms • Final –e replaced with -one. • Longest chain with ketone group is the parent. • Numbering begins at the end nearest the carbonyl carbon. • If ketones is the substituent group, -acyl is used and the name ending –yl is attached.
Practice. . . • CH3CH=CHCH2COCH3 • 4-hexen-2-one • CH3CH2COCH2COCH3 • 2,4-hexanedione • CH3CH2COCH(CH3)2 • 2-methyl-3-pentanone • CH3COCH2CH2CH2COCH2CH3 • 2,6-octanedione
Practice. . . • OHCCH2CH2CH2CHO • Pentanedial • CH3CH2CH(CH3)CH(CH2CH2CH3)COCH3 • 4-methyl-3propyl-2-hexanone • CH3CH=CHCH2CH2CHO • 4-hexenal
Practice. . . • 3-methylbutanal • 3-methyl-3-butenal • 4-chloro-2-pentanone
Carboxylic Acids. . . • Two systems: • Derived from open chain alkanes: • Carboxyl carbon is always C1. • Replace terminal –e of alkane with –oic acid. • -COOH group bonded to a ring, the suffix –carboxylic acid is used. The carboxylic acid carbon is attached to C1 and is not itself numbered in this system.
Practice. . . • CH3CH(CH3)CH2CH2COOH • 4-methylpentanoic acid • HOOCCH2CH(CH2CH3)CH2CH2CH(CH3)CH2COOH • 3-ethyl-6-methyloctanedioic acid
Practice. . . • (CH3)2CHCH2COOH • 3-methylbutanoic acid • CH3CHBrCH2CH2COOH • 4-bromopentanoic acid • CH3CH=CHCH=CHCOOH • 2,4-hexadienoic acid • CH3CH2CH(COOH)CHCH2CH2CH3 • 2-ethylpentanoic acid
COOH Br COOH Practice. . . 3-bromocyclohexanecarboxylic acid 1-cyclopentenecarboxylic acid
Carboxylic Acids. . . • Strong hydrogen bonding has a noticeable effects on boiling points. • Much higher than corresponding alcohols.