Intermolecular Force (IMF)
Intermolecular Force (IMF). Force of attraction between or among molecules. London Dispersion Forces (LDF). Weak force Temporary or induced dipole Exist between all particles Dominant between identical non-polar molecules. Dipole-Dipole. Attraction between oppositely- charged regions

Intermolecular Force (IMF)
E N D
Presentation Transcript
Intermolecular Force (IMF) Force of attraction between or among molecules
London Dispersion Forces (LDF) Weak force Temporary or induced dipole Exist between all particles Dominant between identical non-polar molecules
Dipole-Dipole Attraction between oppositely- charged regions Polar molecules Generally stronger than LDF
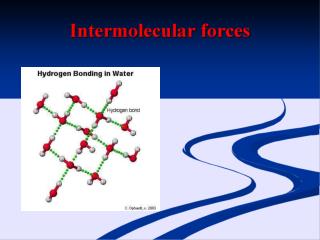
Hydrogen Bond Special type of dipole-dipole Occurs between molecules containing H atom bonded to F, O, N -stronger than ordinary dipole-dipole
H bond>dipole-dipole>LDF polar (N,O,F)polarall molecules -boiling point -melting point -freezing point