I. Intermolecular Forces (Ch. 6, p.189-193)
70 likes | 347 Vues
Ch. 12 - Liquids & Solids. I. Intermolecular Forces (Ch. 6, p.189-193). A. Definition of IMF. Attractive forces between molecules. Much weaker than chemical bonds within molecules. a.k.a. van der Waals forces. B. Types of IMF. B. Types of IMF. London Dispersion Forces.

I. Intermolecular Forces (Ch. 6, p.189-193)
E N D
Presentation Transcript
Ch. 12 - Liquids & Solids I. Intermolecular Forces(Ch. 6, p.189-193) C. Johannesson
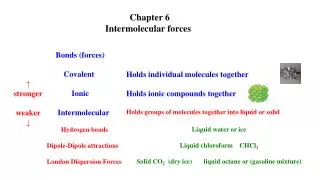
A. Definition of IMF • Attractive forces between molecules. • Much weaker than chemical bonds within molecules. • a.k.a. van der Waals forces C. Johannesson
B. Types of IMF C. Johannesson
B. Types of IMF • London Dispersion Forces View animation online. C. Johannesson
+ - B. Types of IMF • Dipole-Dipole Forces View animation online. C. Johannesson
B. Types of IMF • Hydrogen Bonding C. Johannesson
C. Determining IMF • NCl3 • polar = dispersion, dipole-dipole • CH4 • nonpolar = dispersion • HF • H-F bond = dispersion, dipole-dipole, hydrogen bonding C. Johannesson