Electrolysis
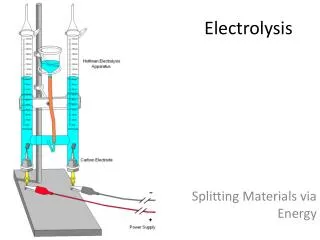
Electrolysis. What is the name of the scientist who investigated the theory of electrolysis in 1830?. Definition of Electrolyis Electrolysis is the conduction of electricity by an electrolyte (usually an ionic compound), when molten or dissolved in water , leading to the

Electrolysis
E N D
Presentation Transcript
Electrolysis What is the name of the scientist who investigated the theory of electrolysis in 1830? Definition of Electrolyis Electrolysis is theconduction of electricity by an electrolyte (usually an ionic compound), when molten or dissolved in water, leading to the decomposition of the electrolyte.
Electrolytes conduct electricity because they contain mobileions. During electrolysis, electrical energy is converted into chemical energy.
Electrolytes Electrolytes is a compound in solution or molten state that conducts electricity with the decomposition at the electrodes as it does so. Electrolytes are usually molten ionic compounds or solutions of ionic salts. Examples: hydrochloric acid, aqueous sodium hydroxide, aqueous sodium chloride
Non-electrolytes Electrolytes Aqueous ethanoic acid Alcohol Dilute nitric acid Sugar solution Pure deionised water Molten lead(II) bromide Copper(II) sulfate solution Pure ethanoic acid Lemon juice Sodium carbonate solution
Why do ionic compounds conduct electricity in molten and aqueous states but not in solid state? In solid state, the ions are held in fixed positions by strong electrostatic forces. In molten or aqueous states, the strong electrostatic forces are weakened/overcome and the ions are now mobile. These mobile ions are able to conduct electricity.
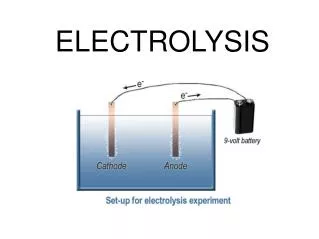
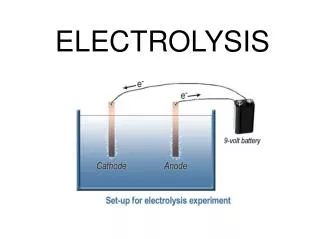
Question for discussion 1. What particles are responsible for electrical conductivity? • Mobile electrons • present in conductors such as metals & graphite • Mobile ions • Present in electrolytes such as dilute HCl or molten NaCl
Question for discussion 2. What substance will remain unchanged when an electric current pass through it? Metals and graphite (elements) 3. What substance will decompose when an electric current pass through it? Electrolytes (E.g molten NaCl)
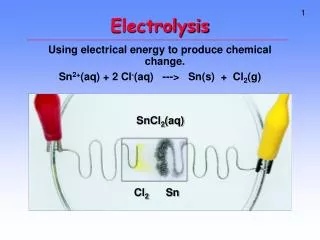
Cathode Anode Anode Electrolysis of Molten lead (II) bromide Electrons flow from anode to cathode Positive electrode Negative electrode ADD ADD
Molten lead(II) bromide contains: Br- Pb2+ And Positive ions, Pb2+, are attracted to negative electrode (cathode) Always remember: Cations are attracted to cathode Negative ions, Br-, are attracted to positive electrode (anode) Always remember: Anions are attracted to anode.
Reactions at the electrodes: Reactions at the anode: 2 Br- (l) Br2 (g) + 2e- Recall, in balancing ionic equations, charge must also be balanced. Charge -2 0 -2 Bromide ions lose electrons to form bromine molecules. Thus in general oxidation occurs at the anode. Remember: An Ox Oxidation Anode Observation at anode: Reddish-brown and pungent gas was observed.
Reaction at cathode, Pb2+(l) Pb(l) + 2e- +2 -2 0 Charge Lead(II) ions gain electrons to form lead atoms. Lead (II) ions are said to be discharged. In general reduction takes place at cathode. Remember: Red Cat Cathode Reduction In the electrolysis of binary molten compounds, a metal is discharged at the cathode; a non-metal is produced at the anode. A binary compound is a compound containing only two elements. Eg NaCl , PbBr2
Eqn at anode Eqn at cathode Molten electrolyte Ions Product Product NaCl Pb2+, O2- PbO 2 O2- (l) O2 (g) +4e- oxygen gas Pb2+ +2 e- Pb lead metal Calcium metal Cal2 Ca2+, l- 2 I- (l) I2(g) +2e- iodine Ca2+ +2 e- Ca Try practice 1 on page 3 of notes Na+, Cl- 2 Cl- (l) Cl2(g) +2e- Chlorine gas (yellowish- Green gas) Na+(l) + e- Na(l) Sodium metal
Question for discussion (NOT in note) During the electrolysis of molten lead(II) oxide, do you expect the lead form to be solid or liquid? Why? What information is needed? Mp of PbO = 888 C Mp of Pb = 327.5 C Lead formed would be in molten/liquid form.
Electrolysis of Solutions Aqueous electrolytes or solutions are mixtures of two electrolytes : the compound and water. Water ionises to a very small extent to give a very small amount of H+ and OH- H2O H+ + OH-
From water From HCl and water 2H+ (aq) - H2(g) +2 e- Electrolysis of Solutions Eg 1 : Electrolysis of concentrated hydrochloric acid Ions present : H+, Cl-, OH- Ions at cathode: H+ Eqn at cathode: Ions at anode: Cl-, OH- There are two anions at anode. Which ion will discharge at anode ?
Factors in deciding which ion is discharged are: • Its position in electrochemical series • Concentration of the ion • Type of electrode
Least readily discharged Hydrogen (from water) (Ions formed by metals above hydrogen in the electrochemical series are not discharged.) Zn2+ Fe2+ Reference point Hydrogen Pb2+ (Depends on concentration) copper silver Most readily discharged Electrochemical Series In an electrochemical series, metals or non-metals are arranged in order of their reactivity. Metallic ions Product at cathode K+ (Group I) Na+ (Group I) Ca2+ (Group II) Mg2+ (Group II) Al3+ (Group III) H+ Cu2+ Ag+
Oxygen (from water) (Sulfate and nitrate ion are not discharged.) SO42- NO3- Least readily discharged O2 if electrolyte is Dilute; respective Halogens if Electrolyte is concentrated Cl- Br- I- OH- oxygen Most readily discharged Non-metallic Ions Product at anode Note: When the non-metallic ions are close in their position in the electrochemical series, order of discharged is influenced by the concentration of anion.
E.g In the electrolysis of concentrated HCl, both OH- & Cl-migrate to the anode. But concentration of Cl- is larger than that of OH- . Hence Cl- is preferentially discharged instead. 2 Cl- (l) Cl2(g) +2e-
Eg 2 Electrolysis of dilute sulfuric acid Ions present in dilute sulfuric acid: H+, OH-, SO42- Ions migrating to cathode: H+ Equation representing reaction at cathode: H+ (aq) H2(g) 2 +2 e- Hydrogen gas Products at cathode: Observation: Effervescence of colourless and odourless gas.
+4e- OH- (aq) H2O(l)+ O2(g) 4 2 Eg 2 Electrolysis of dilute sulfuric acid Ions migrating to anode: OH- SO42- Equation representing reaction at anode: Reason: sulfate ion which is high up in the electrochemical series is very stable. A large amount of energy is required for sulfate to give up its electrons, hence it is not discharged. Thus, hydroxide ions, OH- , is preferentially discharged to give oxygen gas. Product at anode: oxygen gas (and water) Observation: Effervescence of colourless and odourless gas.
The volume of H2 collected during electrolysis of dilute sulfuric acid is twice that of oxygen. Why? Anode: 4OH- 2H2O + O2 + 4e- Cathode: 2H+ + 2e- H2 For 1 mole of oxygen produced at the anode, 4 moles of electrons are released. These 4 moles of electrons will reduce hydrogen ions at the cathode to produce 2 moles of hydrogen gas. Thus volume of hydrogen is twice that of oxygen. Overall equation can be represented as: 2H2O 2H2 + O2 As water molecules are removed/decomposed, concentration of sulfuric acid increases.
Eg 2 Electrolysis of aqueous copper(II) sulfate using graphite electrodes Ions present : H+ Cu2+ SO42- OH- Ions migrating to cathode: H+ Cu2+ Equation representing reaction at cathode: Cu2+ (aq) + 2e- Cu(s) Products at cathode: Copper (brown/pink solid deposited on cathode) [link]
4 2 Eg 2 Electrolysis of aqueous copper (II) sulfate using graphite electrodes Ions migrating to anode: OH- SO42- Equation representing reaction at anode: OH- (aq) H2O(l)+ O2(g) +4e- Products at anode: oxygen and water
Eg 2 Electrolysis of aqueous copper (II) sulfate using graphite electrodes What happens to the electrolyte after some time? The blue electrolyte fades away and eventually becomes colourless. This is due to the reduction of copper (II) ion to copper atoms, leading to a decrease in concentration of copper (II) ions in solution over time. What happens to the pH of the electrolyte? Why? The pH of electrolyte decreases. As hydroxide ions are discharged, concentration of hydroxide ions decreases and is less than that of hydrogen ions, hence making the solution acidic.
Eqn at cathode Eqn at anode Electrolyte Ions Product Product Na+, Cl-, H+, OH-, Na+, Cl-, H+, OH-, Conc NaCl solution Hydrogen gas Hydrogen gas 2 H+(aq) +2e-H2(g) Recall Na+ not dishcarged in aq. form. 2 H+(aq) +2e-H2(g) Recall Na+ not dishcarged in aq. form. 4OH- (aq)2 H2O(l) + O2(g) +4e- Oxygen and water Chlorine gas 2Cl- (aq)Cl2(g) +2e- Dilute aqueous potassium sulfate K+, SO42- H+, OH- Hydrogen gas 4OH- (aq)2 H2O(l) + O2(g) +4e- Oxygen and water 2 H+(aq) +2e-H2(g) Dilute NaCl solution
Reactive electrodes Electrodes Inert electrodes (Electrode has no reaction with the electrolyte. Eg graphite and platinum) Reactive electrodes (Electrode can dissolve in the electrolyte. Eg copper, silver nickel)
Copper electrodes aq. CuSO4 Go to cathode Go to anode Electrolysis of aqueous copper(II) sulfate using copper electrodes Ions present in aqueous copper(II) sulfate: Cu2+, H+ , OH-, SO42- Substances at anode: OH-, SO42- and copper anode. Which substance is most easily oxidised? Copper, a metal, has the greatest tendency to lose electrons and is most easily oxidised to copper (II) ions. Hence at the anode, Cu electrode dissolves. Eqn at anode: Cu (s) Cu2+(aq)+ 2e- Hence anode loses weight and becomes smaller/thinner.
Electrolysis of aqueous copper(II) sulfate using copper electrodes What happens at the cathode? Substances at cathode: H+, Cu2+, Copper cathode Which substance is most easily reduced? Copper(II) ions which are below hydrogen in the electrochemical series has the greatest tendency to gain electrons. At the cathode, brown/pink copper is deposited: Eqn at cathode: Cu2+(aq)+ 2e- Cu (s) Hence cathode gains weight and becomes thicker. The concentration of the electrolyte, copper(II) sulfate, remains unchanged.
What happens to the concentration of copper(II) sulfate? Why? Cu2+(aq)+ 2e- Cu (s) Cathode Cu (s) Cu2+(aq)+ 2e- Anode The concentration of the electrolyte, copper(II) sulfate, remains unchanged. For every mole of copper(II) ion reduced at the cathode, there is equal no. of mole of copper(II) ions being formed in solution due to oxidation of copper at anode.
pure Cu (cathode) - - + + Impure Cu (anode) pure Cu (cathode) Impure Cu (anode) Impurities aq. CuSO4 Application of electrolysis using reactive electrodes - Refining copper The electrolysis of copper(II) sulfate using copper electrodes is used to refine (purify) copper. • The impure Cu is theanode. • A piece of pure Cu is used at the cathode. • Impurities on the anode will fall off. They contain • silver and gold.
Application of electrolysis using reactive electrodes - Refining copper Reaction at anode: Cu (s) Cu2+ (aq)+ 2e- Each copper atom loses 2 electrons to form a copper(II) ion. Hence anode slowly dissolves. It loses weight/reduces in size and becomes thinner. Reaction at cathode: Cu2+ (aq)+ 2e- Cu (s) At the cathode, each copper(II) ion gains 2 electrons to form a copper atom. Hence cathode gains weight and becomes thicker. Concentration of electrolyte remains unchanged.
- + copper anode object at cathode copper (II) sulfate solution Electroplating Electroplating is the process of coating a metallic object with another metal by electrolysis. Electroplating is used for protection against corrosion and for decoration. At the anode, each copper atom loses 2 electrons to form copper(II) ions, hence copper electrode dissolves. Cu(s) Cu2+(aq) + 2e- At the cathode, each copper(II)ion gains 2 electrons and is discharged to form solid copper. Copper metal is plated on the object. Cu2+(aq)+ 2e- Cu (s) Video clip The overall change is for copper to be transferred from the anode to the object.
Electroplating • In electroplating, • the object to plated is the cathode. • the metal used for electroplating is • made the anode. • the electrolyte is a solution of ions of • the metal. Different types of electroplating 1. Tin plating 2. Solder plating 3. Chromium plating 4. Nickel plating 5. Silver / gold plating
After Before Back to slide