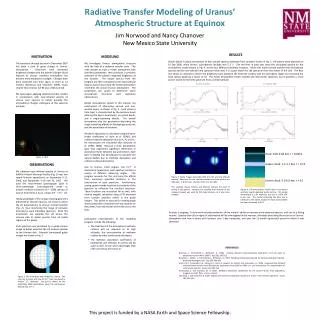
Atmospheric Structure
Atmospheric Structure . Theodore S. Dibble Chemistry Department SUNY-Environmental Science and Forestry Syracuse, NY. Temperature defines regions of the atmosphere. Pressure (mbar). Altitude (km). Pressure and the Barometric Law.

Atmospheric Structure
E N D
Presentation Transcript
Atmospheric Structure Theodore S. Dibble Chemistry Department SUNY-Environmental Science and Forestry Syracuse, NY
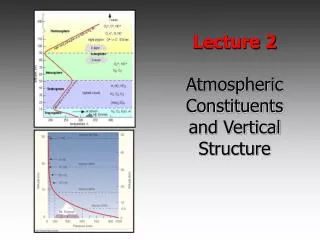
Temperature defines regions of the atmosphere Pressure (mbar) Altitude (km)
Pressure and the Barometric Law P = Poe-Mgh/RTapplies to individual compounds at equilibrium Atmosphere not at equilbrium! Below 130 km, long lived gases have constant mole fraction Pressure behaves as if M =28.8 g/mole (78:21:1 N2:O2:Ar) Exercise: Calculate the partial pressure of O2 at 6 km if PTotal is 0.5 bar.
Atmospheric Composition How can chemistry (reactions) occur?
Units Part per million, etc. by volume = mole fraction 1 ppmv = mole fraction 1 x 10-6 1 ppbv = mole fraction 1 x 10-9 1 pptv = mole fraction 1 x 10-12 Molecules/cm3 (use ideal gas law) 1 atm = 2.5 x 1019 molecules cm-3 at 298 K if PTotal = 1 atm, 1 ppmv = 2.5 x 1013 molecules cm-3 PTotal = 1 Torr 3.2 x 1016 molecules cm-3 At 220 K, 1 Torr = 4.3 x 1016 molecules cm-3
3 months 2 weeks 2 years Mixing Times in Troposphere
Mixing in Troposphere Vertical ground to tropopause in 1 week Horizontal Around a latitude line in weeks Mid-latitude to equator in months Cross equator in 1 year [CO] = 120 ppbv in N.H. and 60 ppbv in S.H. Question: CO has a lifetime of ~2 months Will it be evenly distributed over oceans and remote continental regions within the N.H.?
Key Points • Temperature structure • Most of mass in troposphere • Chemistry competes with transport