VSEPR Theory
VSEPR Theory. Valence Shell Electron Pair Repulsion. VSEPR Theory. Predicts the molecular shape of a bonded molecule Electrons around the central atom arrange themselves as far apart from each other as possible Unshared pairs of electrons (lone pairs) on the central atom repel the most

VSEPR Theory
E N D
Presentation Transcript
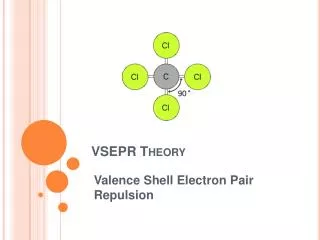
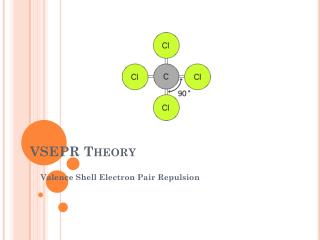
VSEPR Theory Valence Shell Electron Pair Repulsion
VSEPR Theory • Predicts the molecular shape of a bonded molecule • Electrons around the central atom arrange themselves as far apart from each other as possible • Unshared pairs of electrons (lone pairs) on the central atom repel the most • So only look at what is connected to the central atom
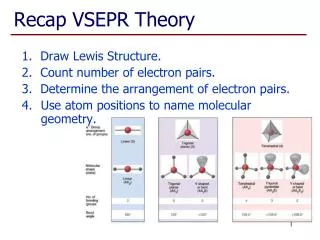
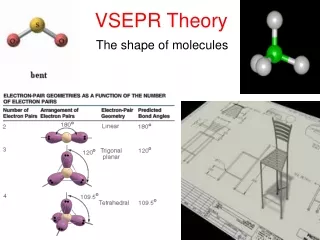
Linear • 2 atoms attached to center atom • 0 unshared pairs (lone pairs) • Bond angle = 180o • Hybrid SP • Molecular –Linear • Ex. BeF2, CO2
Trigonal Planar • 3 atoms attached to center atom • 0 lone pairs • Bond angle = 120o • Hybrid SP2 • Molecular-Trigonal
Tetrahedral • 4 atoms attached to center atom • 0 lone pairs • Bond angle = 109o Hybrid SP3 Molecular-Tetrahedral Ex. CH4
Bent • 2 atoms attached to center atom • 2 lone pairs • Bond angle = 105o Hybrid SP3 Molecular-Bent Ex. Water
Trigonal Pyramidal • 3 atoms attached to center atom • 1 lone pair • Bond angle = 107o Hybrid SP3 Molecular-Pyramidal Ex. NH3
Trigonal Bipyramidal • 5 atoms attached to center atom • 0 lone pairs • Bond angle = • equatorial -> 120o • axial -> 90o • Hybrid SP3D Ex. : PF5
Octahedral • 6 atoms attached to center atom • 0 lone pairs • Bond angle = 90o • Hybrid- SP3D2 • Ex. : SF6
Polar vsnonpolar Covalent bonds means that electrons are being shared between atoms.but the electrons might want to spend more time with one atom than the other.non-polar covalent bonds means that the electrons are shared equally. It's like if you were to share a burger with someone. you eat half, the other person eats half.polar covalent bonds means that the electrons are NOT shared equally. It's like if you were to share a burger with someone and you eat 2/3 of the burger and the other person eats 1/3 of it.
Nonpolar covalent bonds Cl-Cl since the two atoms are identical, they will share electrons equally.Polar covalent bonds: H-O-Hoxygen is an "electron hog" and so the electrons will spend more time with the oxygen than with the hydrogens. unequal sharing.
Using Formal Charge FC. = Valence Electrons – Number of Unshared Electrons – No. of Bonds