Matter and Math
Matter and Math. Chemistry 131 Chapter 1. Matter . Formulas . Physical Properties. Dimensional Analysis. Matter and its Classification.

Matter and Math
E N D
Presentation Transcript
Matter and Math Chemistry 131 Chapter 1
Matter Formulas Physical Properties Dimensional Analysis
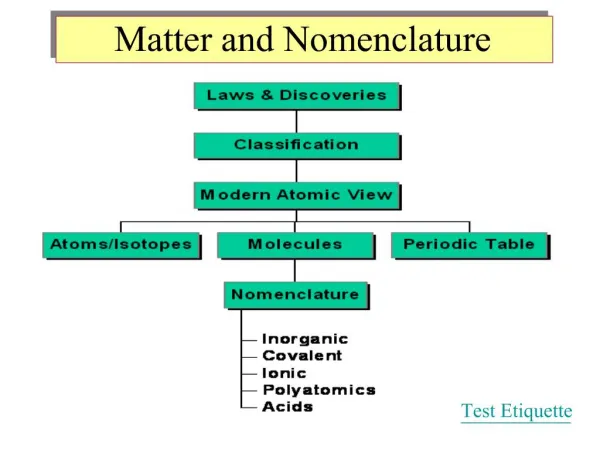
Matter and its Classification This section deals with how we categorize the types of matter into distinct and coherent subgroups. In addition it build a connection map between the different groups.
Can be subdivided into… Matter Everything of substance around you is matter. Whatever has mass is matter. There is a lot more to it than this, but we will start here.
Pure Substances&Mixtures Can be further subdivided into… Matter can be divided into two subgroups, the Pure Substance and the Mixture. Pure substances are those composed of only one element, while mixtures are a combination of at least two elements.
Heterogeneous Mixtures&Solutions Heterogeneous mixtures can be physically separated while solutions cannot. Solutions must be chemically separated.
Can be further subdivided into… Pure Substances&Mixtures Matter can be divided into two subgroups, the Pure Substance and the Mixture. Pure substances are those composed of only one element, while mixtures are a combination of at least two elements.
Can be further subdivided into… Compounds&Elements Compounds are where more than one atom is bonded together in some fashion. Elements are individual atoms.
Molecular Compounds&Ionic Compounds Molecular compounds, or molecules, are those where the atoms are covalently bonded together. The atoms in ionic compounds are held together by ionic bonds.
Compounds&Elements Can be further subdivided into… Compounds are where more than one atom is bonded together in some fashion. Elements are individual atoms.
Metals Metals are those elements on the far right and middle of the periodic table. They hold their outer electrons loosely, have luster, are ductile, malleable, and conductive.
Metalloids Metalloids have properties of both metals and nonmetals, they have luster are typically ductile or malleable to a limited degree and are semi conductive.
Nonmetals Nonmetals are brittle in their solid forms (though they may be in gaseous or liquid forms at room temperature). Typically they are electrical insulators.
Molecular Compounds Compounds Ionic Compounds Pure Substances Metals Matter Elements Metalloids Nonmetals Mixtures Heterogeneous Mixtures Solutions
Chemical Formulas Chemical formulas are the shorthand of chemistry, the language if you will. There are rules to its grammar and syntax that we need to learn and follow as we go through the course.
What is the compound composed of? Take the molecule apart, down to its constituent atoms.
What is there? Now you need to identify the atoms by their elements, in our kits they have standard colors, black for carbon, red for oxygen and white for hydrogen.
Carbon Hydrogen Oxygen
Carbon = 2 Hydrogen = 6 Oxygen = 1
Can we abbreviated anything? Writing out the names of each element every time would be tedious, so each element has an abbreviation, a chemical symbol, to represent it. These symbols may be obvious (O for oxygen) or obscure (W for tungsten, because it used to be Wolfram). These are the symbols used on the Periodic Table.
Carbon = C Hydrogen = H Oxygen = O
Hydrogen = 6 H Carbon = 2 C Oxygen = 1 O
Now add the grammar? The grammar, or syntax, of chemistry is such that elements present as a single atom are represented by their symbol. Elements that are represented by more than one atom are have their atomic symbol followed by a subscript with the number of atoms for that element.
Hydrogen = H6 Carbon = C2 Oxygen = O
Start with the element with the highest number of bonds. Well get into this more later, but the highest number of bonds means the highest number of available electrons and these are the core of how chemistry works.
Hydrogen = H6 Bonds are the holes in this model… little hard to see Carbon = C2 Oxygen = O
Sometimes it isn’t as simple…Let us look at the compound again
Functional Groups… hence…C2H5OH Functional groups are substructures of a compound with specific structure and similar functions. They are written separately in the formulas to emphasize their role in the overall structure and function of the compound.
Physical Properties This section deal with the difference between what it means to undergo a physical versus chemical change.
What is a Physical Property? Physical Properties of matter are those that can be measured or observed without the matter undergoing a chemical change.
Mass Mass is a measure of the amount of matter you have. This is recorded in grams (g) or kilograms (kg). Commonly this is referred to as weight, though technically weight is the mass within a specific gravity field. In other words, the weight may vary, but the mass will not.