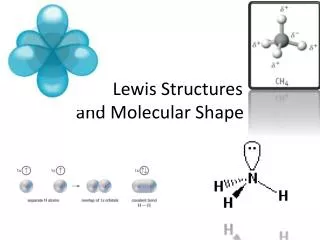
Chapter 8 Molecular Shape
Chapter 8 Molecular Shape. The shape of a molecule can be important in determining its chemical reactions Molecular shape is often very important in the chemistry of living things. Molecular geometry also helps determine the polarity of molecules.

Chapter 8 Molecular Shape
E N D
Presentation Transcript
Chapter 8 Molecular Shape The shape of a molecule can be important in determining its chemical reactions Molecular shape is often very important in the chemistry of living things. Molecular geometry also helps determine the polarity of molecules
High polarity – higher boiling and melting points • CO2 gas is a nonpolar molecule (low boiling point) • H2O is a polar molecule (high melting point)
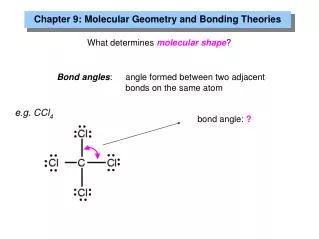
Valence-shell electron pair repulsion theory (VSEPR theory) • Atoms and electron pair groups around an atom repel each other • A molecule will have a geometry that will keep these groups as far apart as possible.
To determine geometry draw an electron dot structure and count the groups around the central atom. (note – a nonbonding electron pair counts as a group.)
Two Groups • Example: CO2 (see electron-dot) • two groups around the central C atom • 180o allows these groups to be as far apart as possible. • Will be linear (180o bond angle).
Three Groups • Example: CH2O see electron-dot structure • Three groups form a triangle around the central C atom. • Called “trigonal planer” geometry (120o bond angle)
4 groups around central atom • Example CH4 (see electron-dot structure) • Form a tetrahedron around the central C atom (109.5o bond angle) • If all 4 groups are atom groups, the geometry is called tetrahedral. • If 3 atom groups and one electron pair, the geometry is called pyramidal. (NH3) • If 2 atom groups and 2 electron pair groups, the geometry is called bent (H2O)
Bond Hybridization • The valence electrons are the s and p electrons. • There is one s orbital and three p orbitals on the outer energy level. • When bonding occurs the orbitals rearrange (hybridize) to create the most stable geometry.
2 groups around atom – sp hybridization • 1 s orbital combines with 1 p orbital to make 2 sp hybrid orbitals. • 3 groups around atom – sp2 hybridization • 1 s orbital combines with 2 p orbitals to make 3 sp2 hybrid orbitals. • 4 groups around atom – sp3 hybridization • 1 s orbital combines with 3 p orbitals to make 4 sp3 hybrid orbitals.
http://www.mhhe.com/physsci/chemistry/animations/chang_7e_esp/bom5s2_6.swfhttp://www.mhhe.com/physsci/chemistry/animations/chang_7e_esp/bom5s2_6.swf
Molecular Polarity • There are two requirements for a molecule to be polar. • 1. The molecule must have polar bonds. (check the electronegativity) • 2. The geometry must allow for a net dipole. • Example – CO2 • Example – H2O