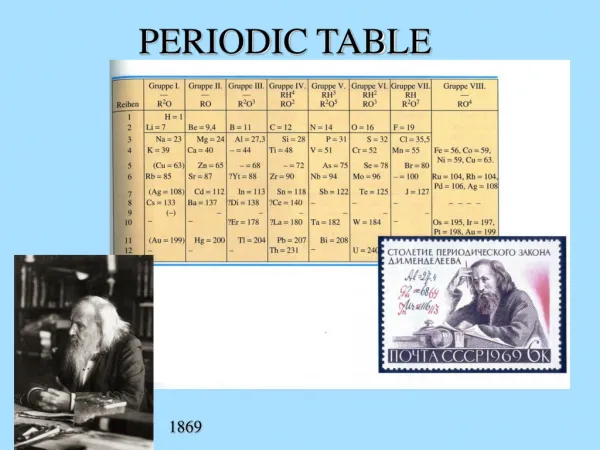
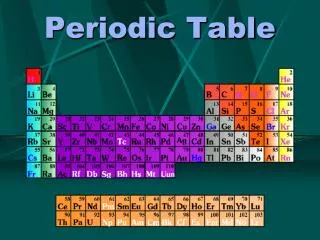
Periodic Table 3
Periodic Table 3. Hydrogen. Hydrogen is diatomic (comes in a pair). Highly reactive (explosive) Hindenburg – famous German zeppelin (airship) that blew up (should have used Helium). Alkali Metals. Group 1 (Lithium, Sodium, Potassium, Rubidium, Cesium, and Francium) Very reactive


Periodic Table 3
E N D
Presentation Transcript
Hydrogen • Hydrogen is diatomic (comes in a pair). • Highly reactive (explosive) • Hindenburg – famous Germanzeppelin (airship) that blew up (should have used Helium).
Alkali Metals • Group 1 (Lithium, Sodium, Potassium, Rubidium, Cesium, and Francium) • Very reactive • Can cut with a butter knife (very soft) • Always combines with something else. • Reacts very violently with water
Alkaline Earth Metals • Group 2 elements • Very reactive metals that tend to combine with non-metals (right side of Periodic table) IMPORTANT TO YOUR HEALTH Ca (Calcium) Mg (Magnesium) deficient = muscle spasms, and has been associated with cardiovascular disease, diabetes, high blood pressure, anxiety disorders, migraines and osteoporosis
Transition Metals • Groups 3-12 • Harder metals that are not as reactive • Jewelry/construction/money
Group 14 • Without carbon, there would not be life on this planet. We humans are carbon based life forms. Plants are carbon based life forms. • Silicon / Germanium (Semiconductors)
Group 15 • Nitrogen (3/4 of our atmosphere) – 75% • Phosphorus (ends of matches) IMPORTANT TO OUR BODIES • Phosphorus is a component of DNA and RNA and an essential element for all living cells. VERY BAD POISON Arsenic – kills you
Halogens • Group 17 (Fluorine, Chlorine, Bromine, Iodine, Astatine, • Volatile (Very reactive) • Does not appear alone (diatomic) – comes in pairs BAD FOR OUR BODY Fluorine /chlorine Gas – poisonous - - causes severe skin burns
Noble Gases • They are noble (don’t fight for electrons…..don’t give up electrons) so they are un-reactive (don’t fight) • Does not come in pairs • Neon (Used in neon signs) • Helium (Used in balloons)
Metalloids / Staircase • A metalloid is an element that has some characteristics of metals and non-metals. • They form a staircase on the Periodic Table (on the border) of metals/non-metals. • Boron (B) Polonium (Po) • Silicon (Si) Tellurium (Te) • Germanium (Ge) Antimony (Sb) • Arsenic (As)
Our BODY • Carbon, the basic unit for organic molecules, comes in second. 99% of the mass of the human body is made up of just six elements: oxygen, carbon, hydrogen, nitrogen, calcium, and phosphorus. • Oxygen (65%) • Carbon (18%) • Hydrogen (10%) • Nitrogen (3%) • Calcium (1.5%) • Phosphorus (1.0%)
Symbols/Names • For the last part of the assignment, you need a Periodic Table. • You look on the Periodic Table to answer the questions. For example, when they give you the symbol Na, you look for Na and give its name (sodium). When they give you its name (sodium), you give its symbol (Na).
Periodic Table- 4 • The Periodic Table shows all the elements. Each element is made up of atoms.
Element Symbol • An element symbol is a short abbreviation…. • Silicon’s symbol is (Si)
Atoms • Atoms have an atomic number, number of protons, number of electrons, number of neutrons, and mass number.
Atomic Number • The atomic number tells you the number of protons and electrons. • When the atomic number is 81, then there are 81 protons and 81 electrons.
Atomic Mass • The atomic mass of an element is the mass of 1 mole of each atom. • If they ask for the mass of 2 moles of atoms…..you times the atomic mass number by 2. Mass of 1 mole of sulfur atoms = 32 Mass of 2 moles of sulfur atoms 2(32) = 64
Neutrons. • The Neutrons of an atom can be found by the mass number minus the number of protons. Oxygen’s mass number = 16 Oxygen’s number of protons = 8 (Mass number) – (Number of protons) = Neutrons (16) - (8) = 8
Inner Transition Metals • The radioactive isotopes on the Periodic Table can be found in the Inner Transitional Metals. • The actinides (bottom row) are radioactive.
Decay • The center (nucleus) of each radioactive isotope is unstable …which means it decays (breaks down) trying to get to a more stable state of being.
Synthetic Radioactive Isotopes • Most of the actinides are synthetic (man-made). • Thorium, Protactinium, and Uranium are the few that are found naturally on earth.
Alpha Particles • Alpha particles are completely stopped by a sheet of paper.
Beta Particles • Beta Particles are stopped by aluminum shielding.
Gamma Particles • Gamma Particles require much thicker dense materials such as a block of lead to stop them.