Chapter 4 Molecules, Compounds, and Chemical Reactions
500 likes | 798 Vues
Chapter 4 Molecules, Compounds, and Chemical Reactions. Molecules cause the behavior of matter. Human body Cooking Cars In all of science history, an exception to this rule has never been found. Molecular Changes. H 2 O 2 hydrogen atoms bonded to 1 oxygen atom

Chapter 4 Molecules, Compounds, and Chemical Reactions
E N D
Presentation Transcript
Molecules cause the behavior of matter. Human body Cooking Cars In all of science history, an exception to this rule has never been found. Molecular Changes
H2O 2 hydrogen atoms bonded to 1 oxygen atom (though this image is likely a mixture of water with other substances) Chemical Compounds and Chemical Formulas
Chemical Formulas • Indicate the elements present in a compound and the relative number of atoms of each • If we change the subscripts in a chemical formula, we no longer represent the same compound. • H2O is water. • H2O2 is hydrogen peroxide.
Concept Check 4.1 • Determine the number of each type of atom in glucose, which has the molecular formula C6H12O6.
Concept Check 4.1 Solution • Each molecule of glucose, C6H12O6 contains: • 6 carbon atoms • 12 hydrogen atoms • 6 oxygen atoms
Ionic and Molecular Compounds • Elements with unstable electron configurations form compounds with other elements to gain stability. • This can be accomplished by the transfer of electrons (ionic compounds) or by the sharing of electrons (molecular compounds).
Ionic Compounds • Contain a metal and one or more nonmetals • Metals and nonmetals are a good chemical match. • Metals lose electrons. • Nonmetals gain electrons. • The result is an ionic bond. • Subscripts represent the ratio of elements in the crystal lattice of the compound.
Soluble ionic compounds dissociate in water to form electrolyte solutions. Such solutions conduct electricity. Sodium Chloride
Molecular Compounds • Contain only nonmetals • Electrons in a bond are shared . . . but not always equally. • The resulting bond is covalent. • Subscripts represent the actual number(s) of each kind of atom in the molecule. • Molecules can be very small or VERY large and VERY complex, such as in protein molecules. hemoglobin molecule
Properties of Molecular Compounds • The bulk properties of molecular compounds depend on the molecules that compose them. • The composition of molecules is responsible for what we observe and experience. • Atoms, shape, structure, and bonds • Small changes in a molecule can dramatically change the properties of the substance.
Concept Check 4.2 • Classify each compound as ionic or molecular. • NaBr • CO2 • MgF2 • CH4
Concept Check 4.2 Solution Compounds formed between a metal and at least one nonmetal are ionic. Compounds formed between nonmetals are molecular. • NaBr: ionic Na (metal) and Br (nonmetal) • SO2: molecular S and O (nonmetals) • MgF2: ionic Mg (metal) and F (nonmetal) • CH4: molecular C and H (nonmetals)
Naming Compounds Ionic • Name begins with the cation (metal) • Followed by the base name of the anion (nonmetal) + ide • NaBr is sodium bromide. • MgBr2 is magnesium bromide. • Names for ionic compounds DO NOT contain prefixes to indicate the number of each type of atom.
Concept Check 4.3 • Name the following ionic compounds • Li2O • CaBr2 • KI
Concept Check 4.3 Solution a) Li2O: lithium + ox(ide) = lithium oxide [note that prefix “di” is not used in front of lithium] b) CaBr2: calcium + brom(ide) = calcium bromide [note that prefix “di” is not used in front of bromide] c) KI: potassium + iod(ide) = potassium iodide
Naming Compounds:Anions and Polyatomic Ions Polyatomic ions are ions that are composed of two or more atoms covalently bonded that behave as a single ionic unit.
Concept Check 4.4 Name the following ionic compounds • Mg(OH)2 • Na2SO4
Concept Check 4.4 Solution Find the name of polyatomic ions on Table 4.2. For naming purposes, the metal atom is treated as it is in other ionic compounds. • Mg(OH)2: magnesium + hydroxide = magnesium hydroxide • Na2SO4: sodium + sulfate = sodium sulfate
Naming Compounds • Calcium carbonate • CaCO3 • Molar mass: 100.09 grams • Melting point: 1339 Celsius • Common in nature • Used in consumer products
Naming Compounds • Molecular • The more metallic element is listed first and is followed by the less metallic element. • Prefixes are used to show the number of atoms of the element that are present. • The mono- prefix is not used on the first element.
Concept Check 4.5 Name the following molecular compounds • CO • N2O5 • SO3
Concept Check 4.5 Solution Name the following molecular compounds • CO: carbon monoxide • Note: no prefix for one carbon and use the “mono” prefix to indicate one oxygen. • N2O5: dinitrogen pentoxide • Note: use the “di” prefix to indicate two nitrogens and the “pent” suffix to indicate five oxygens. • SO3: sulfur trioxide • Note: no prefix for one sulfur and use the “tri” suffix to indicate three oxygens.
Formula Mass • Formula mass of a compound is analogous to the atomic mass of an element. • It is computed by summing the atomic masses of all the atoms in its formula. • Calculating the formula mass of NaCl: Sodium 22.99 amu × 1 = 22.99 amu Chloride 35.45 amu × 1 = 35.45 amu 58.44 amu
Molar Mass • The formula mass of a compound in amu is numerically equivalent to its molar mass in grams per mole (g/mol). • The molar mass of an element is the conversion factor between grams and moles of the element. • It follows then that the molar mass of a compound serves as the conversion factor between grams and moles of that compound. • The mass of one mole of a compound. • Calcium carbonate, CaCO3 Calcium 40.08 grams × 1 mole = 40.08 grams Carbon 12.01 grams × 1 mole = 12.01 grams Oxygen 16.00 grams × 3 moles = 48.00 grams 100.09 grams
Concept Check 4.6 • Calculate the formula mass of the highly toxic gas hydrogen sulfide, H2S.
Concept Check 4.6 Solution Formula mass of H2S = (2 × 1.008 amu) + 32.066 amu = 34.084 amu
Concept Check 4.7 Calculate the molar mass of hydrogen sulfide, H2S.
Concept Check 4.7 Solution Recall from Chapter 3 that the atomic mass of an element in amu is numerically equivalent to its molar mass in g/mol. The same relationship is true for compounds; the formula mass of a compound in amu is numerically equivalent to its molar mass in g/mol. molar mass of H2S = (2 × 1.008 g/mol) + 32.066 g/mol= 34.084g/mol
Chemical Formulas as Conversion Factors • Scientists, manufacturers, farmers, and many others may want to know how much of a particular element is in a certain substance. • How much iron is present in a shipment of iron oxide? • A chemical formula gives us equivalences between the elements in a particular compound and the compound itself.
Concept Check 4.8 Consider a sample containing 5.25 g of H2S. • How many molecules of H2S are contained in this sample? • How many moles of hydrogen atoms are present in the sample. • How many grams of sulfur are in the sample.
Concept Check 4.8 Solution • In Concept Check 4.7, the molar mass of H2S was calculated to be 34.084 g/mol. • The molar mass is the conversion factor from grams to moles of a substance. Avogadro’s number is the conversion factor for moles to number of molecules of a substance.
Concept Check 4.8 Solution • The formula for hydrogen sulfide indicates that for every mole of H2S, there are 2 moles of hydrogen atoms
Concept Check 4.8 Solution • The molar mass of sulfur, 32.066 g/mol is the conversion factor between mass (in grams) and moles.
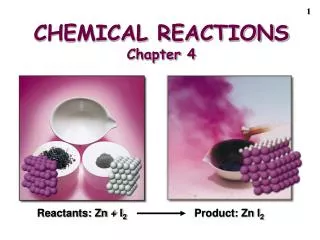
Chemical Reactions • Compounds exist as the result of chemical reactions. • Existing compounds can be transformed by further chemical reactions. • These chemical reactions are represented by chemical equations.
Chemical ReactionsCH4 + 2O2 → CO2 + 2H2O • Reactants, the substances present prior to the reaction, are written on the left side of the equation. • Products, the substances formed by the reaction, are written on the right side of the equation. • The number and types of each atoms in chemical equations must be the same on both sides (balanced) because matter is conserved.
Balancing an Equation • Add coefficients to the reactants, and products make the number of atoms of each type of element on both sides of the equation equal. • This changes the number of atoms and molecules involved in the reaction, but not the types of atoms and molecules.
Balancing Guidelines • If an element occurs in only one compound, balance that element first. • If an element occurs as a free element, balance that element last. • Change only coefficients, never subscripts. • Eliminate fractions; use the lowest whole number ratio of coefficients.
Concept Check 4.9 • Propane, C3H8, is often used in place of methane, CH4, as a fuel for home heating and cooking. Much like methane, it burns in (reacts with) oxygen to produce carbon dioxide (CO2) and water (H2O). Balance the reaction for the burning of propane in oxygen.
Concept Check 4.9 Solution • The unbalanced equation: C3H8 + O2 → CO2 + H2O • First, balance the elements present in only one compound on each side of the equation. C3H8 + O2 → 3CO2 + H2O 3 C’s on each side C3H8 + O2 → 3CO2 + 4H2O 8 H’s on each side • Balance the element present as a free element last. C3H8 + 5O2 → 3CO2 + 4H2O 10 O’s on each side • Final balanced equation: C3H8 + 5O2 → 3CO2 + 4H2O Note: The absence of coefficient in front of a reactant or product implies a “1”.
Reaction Stoichiometry: Chemical Equations as Conversion Factors As was the case with the subscripts within a chemical formula, the coefficients of a chemical equation can be used as conversion factors in chemical calculations, which are mathematical expressions that can answer the question “How much?”
Reaction Stoichiometry: Chemical Equations as Conversion Factors • Coefficients of a chemical equation can be used as conversion factors in chemical calculations. • Campfires as chemical reactions • How much oxygen is needed? • What are the reactants? • What are the products?
Concept Check 4.10 How many moles of nitrogen gas (N2) are required to convert 11.5 moles of hydrogen gas (H2) to ammonia (NH3) according to the following reaction? N2 + 3H2 → 2NH3
Concept Check 4.10 Solution The reaction equation tells us that N2 reacts with H2 in a 1:3 mole ratio which we use as the conversion factor: conversion factor used to convert from moles of H2 available to N2 required
Pizza and Molecules • A balanced chemical equation is like a detailed recipe. • Conversion factors are derived from the equivalences given in the recipe or balanced chemical equation.
Concept Check 4.11 • Suppose the city in which you live burns 2.13 × 108 grams of methane per day (approximate usage for a city with 50,000 inhabitants). How many grams of H2O are produced? The balanced reaction for the combustion of methane is as follows: CH4 + 2O2 → CO2 + 2H2O
Concept Check 4.11 Solution • 2.13 × 108 g of CH4 • CH4 + 2O2 → CO2 + 2H2O conversion factor: grams of CH4 to moles of CH4 conversion factor: moles of CH4 to moles of H2O conversion factor: moles of H2O to grams of H2O
Chapter Summary Molecular Concept • Chemical compounds • Ionic and covalent • Formula mass or molar mass • Chemical reactions • Chemical equations • Reactants and products • Societal Impact • Compounds, natural and manmade, are an important part of our environment. • Chemical reactions keep our society and our bodies going.