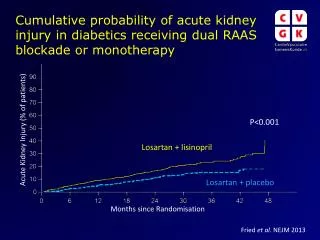
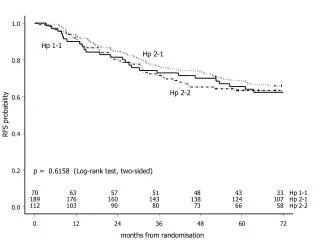
Guidelines for Randomisation in Clinical Study
This document outlines the steps for randomisation in the clinical study via a secure website. Users can access the randomisation system by logging into the provided link. A telephone backup system is available 24 hours a day. Before initiating randomisation, it is crucial to ensure study oximeters are marked as 'available for use' and complete Part A of the Study Entry Form. If allocated oximeters are missing or damaged, users can log in to change them. Proper completion of all eligibility questions is mandatory to proceed with randomisation.

Guidelines for Randomisation in Clinical Study
E N D
Presentation Transcript
Randomisation • Randomisation via secure website • https://rct.npeu.ox.ac.uk/boost/ • Telephone backup system available 24 hours a day
Randomisation • Login: • Select centre • Enter username • Enter password
Randomisation • Before randomising, study oximeters must be assigned as ‘available for use’ • Login to randomisation website • Select Assign new oximeters as ‘Available for use’
Randomisation • Complete Part A of the Study Entry Form before randomising • Login to randomisation website • Select Randomise baby
Randomisation • If the allocated oximeter cannot be found or is broken: • Login to randomisation website • Select Change oximeter (non-routine) If possible, a new oximeter will be allocated
Randomisation • Randomisation cannot proceed if all the eligibility questions are not completed properly