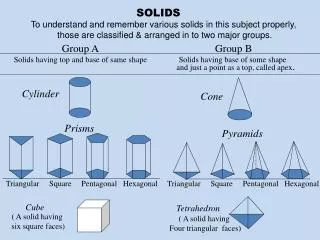
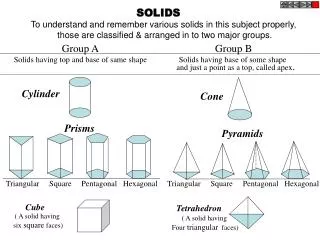
Solids
Solids. Ionic Crystals Strength of attractive forces in ionic crystals is indicated by Coulomb’s law, F = q 1 q 2 /r Forces tend to be very large Crystals are very rigid and brittle. Solids. Molecular Crystals Composed of either atoms of nonmetals or of covalent molecules

Solids
E N D
Presentation Transcript
Solids • Ionic Crystals • Strength of attractive forces in ionic crystals is indicated by Coulomb’s law, • F = q1q2/r • Forces tend to be very large • Crystals are very rigid and brittle
Solids • Molecular Crystals • Composed of either atoms of nonmetals or of covalent molecules • Held together by London forces, dipole-dipole attractions, hydrogen bonding or a mixture of the 3 (weak forces). • Tend to be soft crystals, with low MP
Solids Silicon carbide In diamond- each C atom is covalently bonded to 4 other C atoms in a tetrahedral (sp3) geometry. In graphite, each C atom is covalently bonded to 3 other C atoms in a trigonal planar (sp2) geometry. The extra p e- that isn’t used in bonding holds the sheets weakly together, allowing them to slide. • Network (Covalent) Crystals • Has a lattice structure in which the atoms are covalently bonded to each other • Result is one large molecule with a continuous network of covalent bonds. • Ex. Diamond, silicon dioxide, silicon carbide, graphite
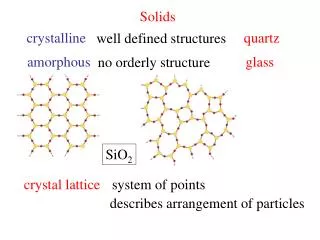
Solids • Amorphous (noncrystalline) solids • Do not have a distinct, sharp MP. They soften gradually over a large temp. range • Examples are glass and many plastics