Solids
Solids. Properties of Solids. Definite shape and volume Particles are close together so attractive forces (bonds and IMFs!) are strong Highly ordered Rigid, incompressible. Two categories of solids.

Solids
E N D
Presentation Transcript
Properties of Solids • Definite shape and volume • Particles are close together so attractive forces (bonds and IMFs!) are strong • Highly ordered • Rigid, incompressible
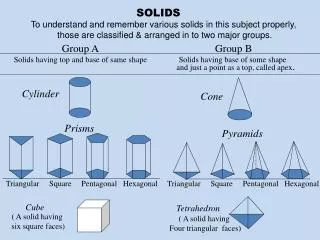
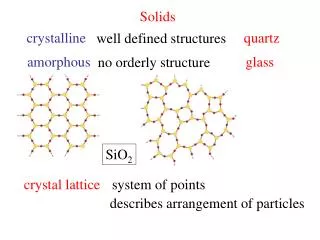
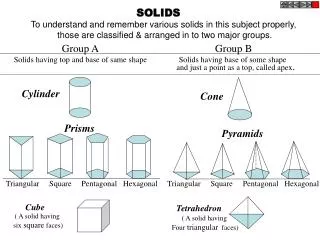
Two categories of solids • Crystalline Solids: have a regular structure in which particles pack in a repeating pattern from one edge of the solid to the other • Amorphous Solids: “solids without form” Have little structure. Includes glass and many plastics
Crystalline Solids • 4 types of crystalline solids based on the types of attractive forces that hold them together. • Molecular solids • Covalent network solids • Ionic solids • Metallic solids
Molecular solids • Composed of molecules held together by IMFs • Relatively soft with low melting and boiling points • Water, sugar, ammonia are all examples
Covalent solids • Form crystals that can be viewed as a “giant” molecule that is held together by an endless number of covalent bonds • Hard, higher melting points and boiling points • Diamond, graphite, quartz are examples
Ionic solids • Held together by the strong force of attraction between oppositely charged ions (cations and anions) • Hard, brittle, high melting and boiling points • Will conduct electricity when in aqueous solutions but not as solids • Salts are examples
Ionic solids • Exist as a 3D repeating pattern called a crystal lattice • Many ionic solids will dissolve in water and dissociate into ions to exist at a lower energy
Ionic Bond Strength • A measure of the attractive forces between the ions • Smaller ions=stronger ionic bonds • Fewer atom ratio=stronger ionic bonds • Evidence comes from melting points! • KCl has a higher melting point than KI because Cl is smaller than I • FeCl2 has a higher melting point than FeCl3 because there are fewer atoms
Metallic Solids • Malleable and ductile • Held together by “metallic bonds” • Have their valence electrons delocalized over many atoms • Good conductors (mobile particles) • “electron sea” like fruit in jello • Examples are gold, aluminum, iron
Why are metal solids malleable while ionic solids are brittle?
Amorphous Solids • No regular geometric pattern • Jumbled up: typically long chains of molecules that get tangled up • Held together by IMFs • Ex: waxes