Unipath 2014
480 likes | 689 Vues
Unipath 2014. Integrated Diagnostic Services for Haematogical Malignancies - Why wouldn’t you? Robin Ireland King’s Integrated Diagnostic Centre Haematological Malignancy and Bone Marrow Failure Syndromes. What is the problem?. (5 th most common malignancy). Size of the problem.

Unipath 2014
E N D
Presentation Transcript
Unipath 2014 Integrated Diagnostic Services for Haematogical Malignancies- Why wouldn’t you?Robin Ireland King’s Integrated Diagnostic Centre Haematological Malignancy and Bone Marrow Failure Syndromes
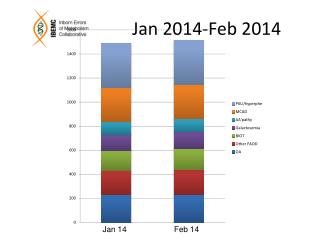
(5th most common malignancy) Size of the problem UK Haematological Malignancy Incidence
Haematological Cancers; estimated annual UK incidence 20,000/yr
12 Major Malignancy Groups (143 subdiagnoses) • Myeloproliferative neoplasms • Myeloid and lymphoid neoplasms with eosinophilia • Myelodysplastic/myeloproliferative neoplasms • Myelodysplastic syndromes • AML • Acute leukaemias of ambiguous lineage • ALL -B and -T • Mature B-neoplasms including all NHL and |Myeloma • Mature -T and -NK neoplasms • Hodgkin lymphoma • Immunodeficiency associated lymphoproliferative disorders. • Histiocytic and dendritic neoplasms.
UK Relative incidences – International geographical variations UK Haematological Malignancy Subtype Data – HMRN
The History of Haematopathology in the UK • The unified concept of haematological malignancy is recent: • Understanding of cellular pathology of the haematopoietic and lymphoreticular systems • UK model dates from the early 1960’s • Pathology subspecialisation • Laboratory Haematology and Histopathology split • Haematologists begin treating leukaemias • Lymphoma and myeloma treated by various specialities • Separation of liquid and solid tissue reporting • Rise of non-morphological techniques • Flow Cytometry • Cytogenetics and Molecular Diagnostics >> FRAGMENTED MODEL
The WHO classification defines diseases as clinico-pathological entities: • Clinical Features • Morphology • Immunohistochemistry • Immunophenotype • Cytogenetics • Molecular genetics
UK NICE Improving Outcomes Guidance: 2003 Diagnosis of Haematological Malignancy • Pathology reviews, audit and trial data suggested significant error rates in diagnosis • Welsh NHS Audit • Accurate diagnosis is increasingly important with changes in treatment and more disease specific directed therapies. • Increasing requirement for cellular prognostic factors and detection of minimal residual disease. • NICE Guidance: ‘Improving Outcomes in Haematological Cancers’ (2003)
USA experience Comparison of referring and tertiary cancer center physician's diagnoses in patients with leukemia. (DeLima M et al. Am J Med. 1998 Mar;104(3):246-51) • 409 patients referred to the M.D. Anderson Leukemia Service • And 100 cases, including 84 of the 409 referred patients for whom the diagnostic outside bone marrow slide had been sent for review. • Overall concordance rate was 73% • 18% discordances were present and considered of major importance (affecting treatment and/or prognosis) • 9% there were minor discordances • Major discordance rates: AML 29% APML 43% MDS 23% CML 5% CLL 2% HCL 57% ALL 19%
Changing Expectation of Diagnostic Services • The Patient and Haemato-oncologist need to have confidence in the accuracy of diagnosis. • The diagnosis needs to be correct and timely. • The morphological opinion of an ‘expert’ is no longer. enough. • Quality is a whole-system process not just NEQAS. • Diagnostic strategies: A systematic approach is essential defining choice and order of testing..
Have raised awareness and improved immunohistological techniques reduced diagnostic errors? 10 years on, is there still a problem? • A recent audit carried out in Greater Manchester (A Norton and R Byers 2008) found the serious and critical error rate to be 15%. These data refer to the diagnosis of lymphoma. • Review was undertaken in a North London Cancer Network. JCO 2011 ……..
Proctor, C. McNamara, M. Rodriguez-Justo, P. Isaacson, and A. Ramsay. Importance of Expert Central Review in the Diagnosis of Lymphoid Malignancies in a Regional Cancer Network, North Central London Lymphoma Whilst error rates have fallen between 2003 and 2008, they are still substantial (13-15%) resulting in minor or major changes in treatment or delay in treatment. Journal of Clinical Oncology April 10, 2011 vol. 29 no. 11 1431-1435
NICE Guidance: Still holds true! • “Improving the consistency and accuracy is probably the single most important aspect of improving outcomes in haematological cancers’’ • “In order to reduced errors, every diagnosis of possible haematological malignancy should be reviewed by specialists” • “Results of tests should be integrated and interpreted by experts” who- -Work with local Clinicians and Pathologists (the final Quality check) to provide a specialised service at network level’
What does this mean in practice? How can it be provided?
Management of patients should be based on sound and comprehensive diagnostic information: An integrated diagnostic process is required Best achieved by co-locating all specialist haematopatholology diagnostic services in a single laboratory. Integration of results into a single final report. Key NICE Recommendations
WHAT IS NEEDED TO MAKE IT WORK? • Agree national model • Identified integrated diagnostic centres • Process of change to implement new model on historical practices and referral practices
An integrated Haematopatholology service is not just an integrated report Multiple levels of integration required. • Integrated approach to the diagnostic process: • Cluster of laboratories and ‘-ologies in an effective diagnostic network • Breakdown historical barriers between laboratories and sub-specialities • Single managerial control, appropriate staffing, organisational and operational structures • Single point of reception for all samples • Design of systematic diagnostic protocols • Integrated IT system • Integrated Final Report combining results from all data sources • Close interface between clinical and laboratory services
Integrated diagnostic processes • Are the best guarantee of diagnostic accuracy based on: • WHO Classification • Systematic diagnostic protocols • Diagnostic protocols designed to give independent checks of diagnosis • Concordance of results across specimen types and diagnostic platforms
A systematic approach is essential • INPUTS: WHO • Clinical Features • Morphology • Immunohistochemistry • Immunophenotype • Cytogenetics • Molecular genetics Accurate Diagnosis Depends on the Integration of Multiple Diagnostic Modalities Immunophenotyping Cytochemistry OUTPUT: The Integrated Report: New diagnoses ICD-O/3 coded Coded comments on: Prognostic factors Minimal Residual Disease monitoring General descriptors Chimerism and Transplant monitoring Free text comments Cytogenetics Morphology Molecular FISH
CURRENT KING’S SERVICES • Fully Integrated Diagnostics and Reporting since October 2007 • Staffing: ~25 staff (Excluding Consultants) • Haematological cytomorphology • Histopathology/Immunohistochemistry/Cytopathology • Flow Cytometry • Cytogenetics/FISH • Molecular Diagnostics • IT and other infrastructures.
Adult Population ~5.3m ~12,000 referrals and ~22,000 tests King’s Haematological Malignancy Diagnostic Centre • All haematological malignancies: Acute, Chronic, Myeloid, Lymphoid, Plasma Cell, Histiocytic, Dendritic. • 3 Cancer networks • 17 Hospitals • Specimens: • Peripheral blood • Marrow Aspirate and Trephines • Tissue biopsies; Ln’s, extranodal, skin, lung etc • CSF/Vitreous Humour /Ascites/Pleural/ Pericardial fluids.
WHAT IS NEEDED TO MAKE IT WORK? • Laboratory services and staff need to be robust and flexible: • Rapid development/implementation of new technologies • Challenges traditional technical/-ology borders • Platform of technology research feeding diagnostic service • Changes in skill mix and cross-cover. • Capital investment capability
Integrated Haematopathology Services: - Organisation • A single Centre with overall responsibility: • Operational, managerial and financial • Identified laboratories and investigational modalities. • Minimum catchment population • May provide services to more than one Network • Management and organisational accountability • Single Head of Service • Defined role and responsibilities including: • Design of algorithms • Resource utilisation • Links with clinicians • Specimen(s) sent to a single reception point • Identified optimal samples, methods of handling and transport • Speedy transfer of samples to minimise delays of diagnosis • Prevent omission of key tests and over-investigation
IMPROVED PROCESSES Referred Samples IHMDC Reception NHS No. or Lifelong and Event Specific ID Finite cell samples – use to maximum diagnostic benefit in shortest possible time. Most samples come with diagnosis of ‘cytopenia or splenomegaly ? Cause. Even if given a diagnosis, often incorrect!! Cytogenetics /FISH Result Molecular Genetics Result Histopathology /Cytology Result Morphology/ Immunopheno -typing Result Initial review determines downstream investigations Consultant Worklist Trafficlight results status Integrated Results ICD-O/3 Codes Interpretive comments Interim/Final Report Authorised Paper and Electronic issue
KCH Sample flows 2013 – Centralised model Single Reception > Testing > Integrated Report
WHAT IS NEEDED TO MAKE IT WORK? -Information Technology Systems • I.T. software is the ‘glue’ that makes it a workable and effective system: • Patient, clinical and sample details recorded. • Investigative protocols: • Systematic, sequential test ordering protocols • Worksheet generation • A single integrated report linking all results. • Communications with users: • Single point direct access • Use of e-mail alerts and electronic return of results. • Quality assurance and audit
Referred Samples HMDC Reception NHS No. or Lifelong and Event Specific ID Cytogenetics /FISH Result Molecular Genetics Result Histopathology /Cytology Result Morphology/ Immunopheno -typing Result Initial review determines downstream investigations Consultant Worklist Trafficlight results status Final check of result quality, internal consistency, concordance or DISCORDANCE of results Integrated Results ICD-O/3 Codes Interpretive comments Interim/Final Report Authorised Paper and Electronic issue
WHAT IS NEEDED TO MAKE IT WORK? - Quality Assurance • Quality and governance built into the system, facilities and processes. • Standard Operating Protocols for order and choice of tests. • Accuracy of reporting: • Double reporting/authorisation • Result concordance • CPA, NEQAS, EQA • Monitoring of reporting times • Monitoring of resource utilisation and efficiency.
Supporting structures • Education and training: • Technical/Scientific • Postgraduate: Trainee Registrars all rotate into HMDC • Consultants
Key Requirements: R&D Infrastructure • Diagnostic Techniques are changing rapidly: • Relative decline of morphology (remains an important triage step) • Multicolour flow techniques (8 – 10 colour). • Gene expression profiling, Deep Sequencing capabilities, SNP-A karyotyping, miRNA’s, whole exome sequencing etc. • Diagnostic services underpinned by large R&D infrastructure and capability. • New skills required – Bioinformatics and mathematical algorithms • Clinical needs are evolving: • New treatments and monitoring should be seen as a package • Eg CML and BCR-ABL/ABL ratios • Centre for Clinical Trials in Haematological Malignancies. • Now incorporated into Current trials – e.g. REMoDL-B…….
Concerns – Role of District Hospital Haematologists • Haematologists should still report their own aspirate/trephine slides. • Local Haematologists do not usually undertake the specialist tests (Immunophenotyping, Cytogenetics, FISH, Molecular Genetics) so for the majority this is not a real change. • If there are haematologists/histopathologists who have a mainly Haem-Oncology laboratory role they could/should be incorporated into the provision of services centrally: • Cancer Pathology Commissioners and Hospitals must be brought into the discussions so that relevant consultants can participate in diagnostic service provision. • Will see more and access a wider range of diagnostic technologies.
Concerns - Deskilling • Clinicians can and should still combine their clinical and laboratory skills. • They should continue to report bone marrows; this is an important quality assurance step where treatment decisions are made. • IOG clarification is not about morphology skills of haematologists (which they are encouraged to maintain) but is about the proper integrated diagnostic process of specialist testing - core to the NICE guidance.
The case for a centralised model Why would you leave a distributed (fragmented) service model? • Difficult to implement modern, complex, integrated diagnostic approaches. • Complex transport arrangements, delays, risks of sample loss. • Many cases come without a firm or correct diagnosis: • Slow/difficult communication between labs to change investigations. • Slow to switch samples between labs; sample degradation and lengthened delays. • Inability to discuss results from discordant cases with all slides/ results/people simultaneously. • Inhibits cohesion, communication, coordination and teamwork • Inefficient utilisation of scarce staff resources and skills • Compromises critical mass for development, learning and research • No multi-tasking and multi-skilling = Inefficient labour force • Does not facilitate service delivery, teaching, tissue banking or research.
Patient/Clinician/Organisation Who wants? • A slow result with a 10 – 25% error rate? • Laboratory reduplication with high capital costs? • Inefficient use of resources/high revenue costs? • Non-robust staffing? • No multiskilling? • Slow implementation of technologies? • Poor training? • Defunct research? - - especially molecular capability.
THANK YOU HAEMATOLOGISTS Prof G Mufti Dr R Ireland Dr V Tindell Dr D Yallop Dr S Kassam HISTOPATH J Salisbury S Pomplun C Shah H Abu-Arqoub J Morehead CYTOGENETICS B Czepulkowski L Brown P Ford H Gilbert T Kontou D Ladon V Simonian D Jijon FLOW CYTOMETRY Tim Milne Anne Etches Melissa Bullard Debbie D’Costa MOLECULAR Nic Lea A Kizilors N Folarin S Best J Anwar RECEPTION C Sulzdorf R Connell