1st and 2nd Order Reactions
80 likes | 373 Vues
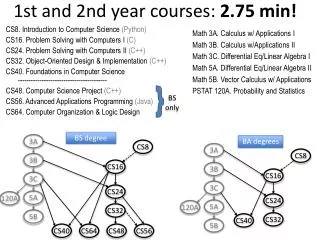
1st and 2nd Order Reactions. Rate laws can be converted into formulas to help us find the concentration of reactants at various time during a reaction. First Order Reactions Depends on only one reactant Rate can be integrated to create

1st and 2nd Order Reactions
E N D
Presentation Transcript
1st and 2nd Order Reactions Rate laws can be converted into formulas to help us find the concentration of reactants at various time during a reaction • First Order Reactions • Depends on only one reactant • Rate can be integrated to create • ln[A]t – ln[A]0 = -k t • [A]t = concen of A at some time t • [A]t = concen of A originally • k = rate constant • t = time (must have same units as k!)
Example A first order reaction causes a protein in our bodies to break down. The rate constant at body temperature is 4.6 x 10-3 s-1. How long does it take for our body to cause the concentration of the protein to change from the normal amount (4.7 x 10-5 M) to the lethal level of 1.9 x 10-5 M? ln[A]t – ln[A]0 = -k t ln(1.9 x 10-5) - ln(4.7 x 10-5) = -4.6 x 10-3 (t) -10.87 – (-9.96) = -4.6 x 10-3 (t) t = 197.8 sec
Example The decomposition of N2O5 is a first order reaction. At 10oC, the rate constant is 5.9 x 10-4 s-1. If the initial concentration of N2O5 is 0.0250 M, how much is left after 25 seconds? ln[A]t – ln[A]0 = -k t ln(x) - ln(0.0250) = -5.9 x 10-4 (25) ln(x) – (-3.689) = -1.475 x 10-2 ln(x) +3.689 = -1.475 x 10-2 ln(x) = -3.703 e(ln(x)) = e-3.703 X = 0.0246 M How do you remove ln? Use ex (ln 5 = 1.609 So e1.609= 5)
B. Half life of first order reactions First order rates can be used ot calculate ½ life of a chemical reaction Half life = time it takes for half of the reactant to be consumed. Does not matter what the initial concentration is, only what k is. Example If the rate constant for a 1st order reaction is 1.5 x 10-3s-1, what is the half life? (You can use any starting concentration!) ln[A]t – ln[A]0 = -k t ln(0.5) – ln(1) = -1.5 x 10-3 (t) -0.693 – 0 = -1.5 x 10-3 (t) t = 462 sec
C. Second order reactions Either 2 of the same reactants hit (2A) or 2 different molecules hit (A + B) We will only look at 2A type Integrate rate= k[A]2 and you get 1 - 1 = k t [A]t [A]o Example – the creation of a hormone is second order, combining 2 molecules of protein C. If the rate constant is 3.6 x 102 M-1s-1, how much C is left after 2.80 seconds if we start with a concentration of 2.9 x 10-4 M of C?
1 - 1 = k t [A]t [A]o 1 – _ 1 . = (3.6 x 102)(2.80) Ct 2.9 x 10-4 __1__ - 3448.3 = 1008 Ct __1__ = 4456.3 Ct Ct = 2.24 x 10-4 M
D. Half life for 2nd order reactions This DOES vary with initial concentration. Must know the starting concentration to determine the half life. Example – What is the half life of a second order reaction with a rate constant of 2.5 M-1s-1 and an initial concentration of 1.0 M? 1 - 1 = k t [A]t [A]o _1_ - _1_ = 2.5 (t) 0.5 1 2 – 1 = 2.5 (t) t = 0.4 sec
Example – Using the same reaction, what is the ½ life if the initial concentration is 2.0 M? 1 - 1 = k t [A]t [A]o _1_ - _1_ = 2.5 (t) 1 2 1 – 0.5 = 2.5 (t) t = 0.2 sec Notice – Higher initial concentration, faster half life!