Cancer stem cells
Cancer stem cells. IOSI Journal Club Giulia Poretti January 19, 2007. stem cells (sc). Stem cells → progenitor cells → mature cells. SELF-RENEWAL i.e. replenish the repertoire of identical stem cell DIFFERENTIATION

Cancer stem cells
E N D
Presentation Transcript
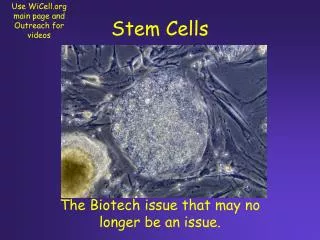
Cancer stem cells IOSI Journal Club Giulia Poretti January 19, 2007
stem cells (sc) Stem cells → progenitor cells → mature cells • SELF-RENEWAL i.e. replenish the repertoire of identical stem cell • DIFFERENTIATION i.e. create a heterogeneous progeny differentiating to mature cells • EXTRAORDINARY PROLIFERATION POTENTIAL HOMEOSTATIC CONTROLaccording to the influence of microenvironment. Modified from Clarke MF et al. Cell. 2006;124:1111-1115
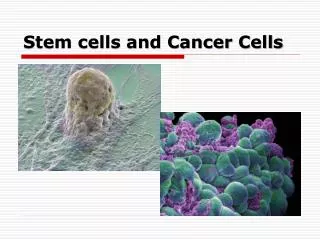
cancer stem cells (csc) Minority of cancer cells with tumorigenic potential NORMAL • SELF-RENEWAL • DIFFERENTIATION • PROLIFERATIVE ABILITY ABERRANT REGULATION TUMORAL Modified from Bjerkvig R et al. Nat Rev Cancer. 2005;5:899-904
stem cells: identifying properties • SELF-RENEWAL • DIFFERENTIATION • EXTENSIVE PROLIFERATION POTENTIAL • Are the minority subpopulation in a given tissue • Mainly appear to be in a quiescent cell-cycle state • long-lived cells giving rise to short-lived, differentiated cells • Highly influenced by signals form their microenvironment • Characterized by specific surface markers
Therapeutic implications • Resistance to treatment → absence of the targeted biological property (imatinib mesylate in CML) → quiescent state → expression of efflux proteins protecting vs xenobiotic toxins • Relapse • Metastasis Strategies to target cancer stem cells: • Immunotherapy against stem-cell-specific markers • Combination of treatment vs tumor burden and treatment vs cancer stem cells • Therapies promoting differentiation of cancer stem cells
Assays in stem cell research Surrogate in vitro and in vivo studies • Clonogenic assays • Repopulation experiments in immunodeficient mice strains STEM CELLS • 1960s: transplantation experiments in immunodeficient mice →very small population of cells responsible for reconstitution →surface marker phenotype negative for lineage-specific antigen CANCER STEM CELLS • 1990s: AML cells transplanted in immunodeficient mice →cells able to sustain tumor growth are a minority subpopulation →reconstitution of the phenotypic heterogeneity of donor tumor
Brain tumor:„Neurosphere“ assay • Cell culture system for normal neural stem cells → long-term self-renewing → multi-lineage-differentiating • Galli R et al. Cancer Res. 2004;64:7011-7021: isolation and serial propagation of „cancer neurospheres“ → long-term self-renewing → multi-lineage-differentiating → in vivo tumorigenicity • Singh SK et al. Nature. 2004;432:396-401: Cell surface marker CD133 identifies glioma stem cells
Cancer stem cells models • Acute myelogenous leukemia: [CD34+,CD38-] • Breast Cancer: [CD44+, CD24-/low] • Brain tumor: [CD133+] • Prostate cancer: [CD44+] • Colon cancer: [CD133+]
Cancer stem cells models • Glioma stem cells are identified by CD133+ cell-surface marker • Glioma CD133+ cells are resistant to radiation • Radioresistance due to more efficient activation of DNA damage checkpoint • Proof of principle: radioresistance of CD133+ glioma stem cells can be reversed with inhibitor of DNA damage checkpoint • Biological explanation of the long-term failure of radiation therapy: tumorigenic subpopulation of CD133+ glioma cells is not eliminated
Experimental models in vitro models (ex vivo ) • Cultured cell from human glioma xenograft: D456MG D54MG • Patient glioblastoma samples in vivo models • Human xenograft models in immunocompromised mice
in vivo CD133+ enrichment after radiation • Glioma xenograft D456MG: →no significant difference between sc and ic →enriched CD133+ population 48h after radiation (3-5x)
in vitro CD133+ enrichment after radiation • Cultures from human glioma xenograft (D54MG): →48h after radiation: 3x enrichment • Patient glioblastoma samples:
in vitro CD133+ enrichment after radiation • CD133+ and CD133- cells derived from patient glioblastoma sample: → separately dye-labeled CD133+ (green) CD133- (red) → mixed (5%CD133+)
CD133+ enrichment due to clone selection CD133+ expression is not induced by irradiation
Irradiation effects at molecular level DNA damage (alkaline comet assay): CD133+ cells repaired the DNA damage more efficiently than CD133-
Irradiation effects at molecular level Early DNA damage checkpoint responses: Early DNA damage checkpoint responses (phosphorylation) checked before treatment and after 1h. Higher amount of phosphorylated proteins in CD133+.
Radioresistance at molecular level in vitro irradiation in vivo irradiation Activation of cleaved caspase-3 (apoptosis) assessed after 24h
Radioresistance at molecular level in vitro irradiation Activation of apoptosis assessed after 20h
Radioresistance:proof of principle at cellular level Cell survival as assessed by colony formation assay
Radioresistance:proof of principle in vivo DNA repair machinery induced by DNA damage is as promizing drug target to overcome radioresistance.
tumorigenic potential proportional to CD133+ • Increased CD133+ cell fractions dose-dependently • decreased tumor latency • increased tumor growth and vascularisation
serial propagation of tumor (secondary tumor formation) Tumor cells derived from irradiated xenografts are enriched in CD133+ tumor cells and show increased tumorigenic potential when xenotransplanted in immunocompromised mice
serial propagation of tumor with selected CD133+ CD133+ cells derived from xenografts are patient sample show tumorigenic potential independently of prior irradiation.
in vivo tumorigenic potentialof selected CD133+ tumor cells in vitro irradiation CD133+ cells (104) from patient sample or xenograft transplanted into brains of immunocompromised mice. Brain observed at appearence of neurological signs or after 8 weeks. D456MG CD133- (2 x 106) formed small tumors in 2 out of 5 xenotransplanted in immunocompromised mice.
„Cancer neurospheres“ assay Purified CD133+ tumor cells from glioma xenografts (D456MG) and patient samples (T3379, T3317) form neurospheres.
Expression of specific surface markers • Multi-lineage differentiation ability
Stem cell-specific markers Identified on neurospheres formed from CD133+ tumor cells from glioma xenografts (D456MG) and patient samples (T3379) by immunofluorescence.
Markers of differentiated cells: in vitro in vitro irradiation
Markers of differentiated cells: in vivo Immunofluorescent staining of frozen sections of tumors generated by CD133+ (source not specified)
Concluding remarks • Glioma cell lines D456MG and D54MG are p53 wild-type • Radiation on individual cells ex vivo: → absence of specific microenvironment • Lack of conservation in the experimental models adopted for the different assays
Haematoxylin: blue staining of the nuclues Eosin: pink staininig of cytoplasm
Remarks • Glioma cell lines D456MG and D54MG are p53 wild-type • Radiation on individual cells ex vivo: → absence of specific microenvironment • CD133+ glioma stem cells treated with ChK inhibitor DBH were not xenotransplanted to evaluate tumorigenicity