Protein Structure
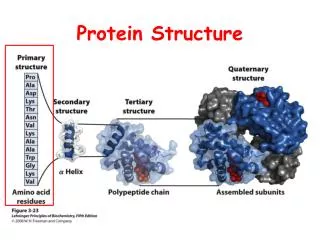
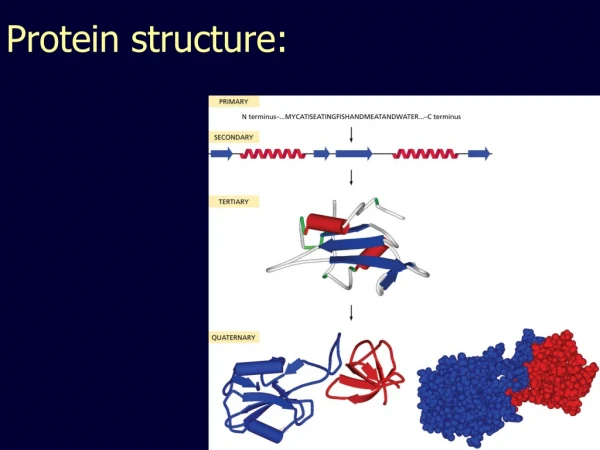
Protein Structure. September 4, 2008. Basics of Protein Structure. Primary structure: sequence Secondary structure: α -helix, -sheet, -strand, loop Supersecondary structure, motifs Tertiary structure:

Protein Structure
E N D
Presentation Transcript
Protein Structure September 4, 2008
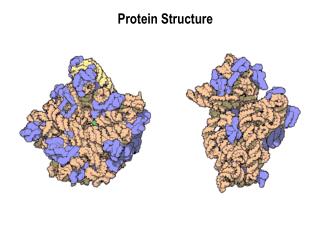
Basics of Protein Structure • Primary structure: sequence • Secondary structure: • α-helix, -sheet, -strand, loop • Supersecondary structure, motifs • Tertiary structure: • folding into functional domains with ordered structure composed of secondary structure elements • Quaternary structure • Complexes of monomers to form active structures
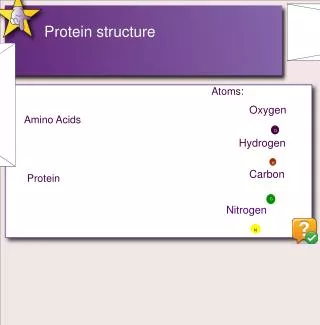
Amino acid categories • Aliphatic • Valine, Alanine, Leucine and Isoleucine • Aromatic • Phenylalanine, Tyrosine and Tryptophan • Charged • Aspartic, Glutamic, Histidine, Lysine, Arginine • Polar • Serine, Threonine, Cysteine, Methionine, Asparagine, Glutamine • Odd couple • Glycine, Proline
Charged Residues • Side-chains are charged under physiological conditions • Acid are negatively charged • Basic are positively charged
w Backbone torsion angles • f phi y psi • omega 180° trans 0° cis
Other chemical bonds • Disulfide bonds or bridges • Formed by oxidation of thiol groups of two cysteines • Form a bond about 2Å in length. • Predominant feature in many small proteins • H-bonds not truly covalent • Dipolar attraction between O and H • Complex geometry wrt distances and angles
Side Chain Torsion Angles The side chain torsion angles are named c1(chi1), c2(chi2), c3 (chi3), etc., as shown below for lysine.
Hydrophilic or hydrophobic…? • Virtually all soluble proteins feature a hydrophobic core surrounded by a hydrophilic surface • Peptide backbone is inherently polar • Neutralize potential H-donors & acceptors using ordered secondary structure
Alpha helix • H-bonds between N-H and C=O groups in polypeptide backbone • Compact structure • 3.6 residues • Pitch: 5.6Å/turn • Rise: 1.5Å/residue • Polar/hydrophilic residues on 1 face with nonpolar or hydrophobic residues on other face
Secondary Structure: -helix • 3.6 residues / turn • Axial dipole moment • Not Proline & Glycine • Protein surfaces
Beta-sheet • Extended structure • Side-chains project alternately up or down • Amphipathic is solvent exposed • polar residues on one side and non-polar on other side
Secondary Structure: -sheets • Parallel or antiparallel • Alternating side-chains • No mixing • Loops often have polar amino acids
Supersecondary structures • Also called motifs • Simple combinations of secondary structures
corners are observed to have a right-handed twist when viewed from the concave side
Tertiary Structure • Combinations of motifs to form domains • Three main classes • All alpha • Alpha/beta • All beta
Alpha Domain Structures • Four helix bundle • Globin fold
Alpha/beta • TIM barrel • Rossmann fold • Horseshoe fold