Understanding Concentration, Molarity, and Dilutions in Solutions
90 likes | 235 Vues
This document explores the concepts of concentration, molarity, and dilution of solutions in chemistry. It defines a mole as a unit for expressing the concentration of solutions, differentiating between concentrated and dilute solutions. Molarity is explained with formulas for calculating the amount of solute in liters of solution. Key examples illustrate these calculations, including diluting solutions and finding percentage concentrations. Overall, it serves as a guide for understanding how to prepare and analyze solutions in a laboratory setting.

Understanding Concentration, Molarity, and Dilutions in Solutions
E N D
Presentation Transcript
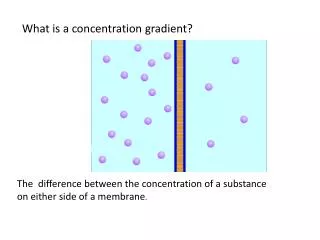
Concentration of Sol’n16-2 The amount of solute in a given amount of solvent or solution
THE MOLE • The mole is used to express the concentration of a solution. • Concentrated sol’n contain large amounts of solute and small amnt of solvent • Dilute sol’n contain small amnts of solute and large amnts of solvent.
Molarity • If defined in terms of volume of soln, depends on T. • Molarity = moles of solute liters of solution • M = mol L
Example #1 • If a manufacturer prepares a soln from 10.0g of NaOH dissolved in enough solvent to make .10L of soln. What is the M of the cleaner? • 10gNaOH x 1mol NaOH = 2.5 mol/L .10L 40g NaOH • 2.5M
Mole Fraction • See Practice Worksheet #1
Making Dilutions • Diluting a solution reduces the number of moles of solute per unit volume, but it does not change the total number of moles of solute. • Moles of solute B4 dl = mols of solute after dl • M1V1= M2V2
Example #3 • How many mL of aqueous 2.0M MgSO4 solution must be diluted with water to prepare 100mL of aq. 0.40 M MgSO4? • M1 = 2.0M • M2= .40M • V1 = ??? • V2 = 100mL
Percent Solutions • Conc. of solution in % expresses the ration of the volume of solute to the volume of solution. • When both the solute and the solvent are liquids • Ex. 91% alcohol contains 91 mL of isopropyl alcohol (2-propanol) with enough water to make 100 mL of solution • %by Volume = volume of solute x 100 volume of soln
Example #4 • What is the percent volume of ethanol in the final solution when 85 mL of ethanol is diluted to a volume of 250mL with water? • A bottle of hydrogen peroxide is labeled 3%. How many mL of H2O2 are in a 400mL bottle of this solution?