Homeostasis
Homeostasis . *All living things must maintain a balance regardless of internal and external conditions. Homeostasis. The process of maintaining equilibrium in the cell’s internal environment, a resistance to change. *This is similar to a thermostat in your home.

Homeostasis
E N D
Presentation Transcript
Homeostasis *All living things must maintain a balance regardless of internal and external conditions
Homeostasis • The process of maintaining equilibrium in the cell’s internal environment, a resistance to change *This is similar to a thermostat in your home
Important terms to know: • Solute: substance being dissolved in another substance (often the lesser quantity) • Solvent: substance that dissolves another substance Example: Kool-Aid, Which is the solute and which the solvent?
Mixture vs. Compound • Compound: cannot be easily separated by physical methods • The chemical/physical properties are totally different than the properties of the parts
NaCl • Na-very reactive, whitish, Cl-poisonous, green gas • We know this compound as table salt. • H2O2 • H2- explosive gas, O2-flammable gas • We know this compound as hydrogen peroxide.
Mixture: can be easily separated by physical methods • The chemical/physical properties remain the same
Three types of mixtures: 1. Solution: two or more substances in a single phase, does not settle out, it is mixedevenly • *solute molecules are smaller than in the solvent • ex. Sugarwater
Three types of mixtures: 2. Suspension: solute larger than solvent will settle out over time, unless the mixture is constantly stirred or agitated • ex. muddy water-mud settles when not stirred, particles can be separated
Three types of mixtures: 3. Colloid: particles of solute and solvent are mainly the same size, gel-like • small particles remain suspended • ex. types of emulsion-oils, waxes • smoke, fog-liquid in gas
Each of these mixtures is in our body: • Solution: saltwater • Suspension: blood • Colloid: Cytoplasm
Major part of cells that maintain homeostasis: • 1. Plasma Membrane: • a. Function: boundary between cell and its environment, allows steady supply of nutrients to come into the cell and excess wastes out-too much can be harmful • Cells need nutrients-glucose, aminoacids, lipids
b. Selective permeability: allows some molecules in and keeps others out • c. Structure: • phospholipidbilayer: two layers of phospholipids back to back • glycerol, fatty acids, phosphate • Nonpolar: phospholipids, no charge • Polar: phosphate can dissolve other substances • (“like dissolves like”…so nonpolar can only dissolve nonpolar)
Water is polar, so it can interact with phosphate (facing outside of the cell) • Fatty acids avoid water (face middle of cell membrane) • Hydrophobic: water fearing • Hydrophilic: water loving
d. Fluid mosaic model: membrane is flexible, proteins create a pattern on the membrane surface • Transport proteins: allow needed material or wastes to move through the membrane, movement of substances in a mixture
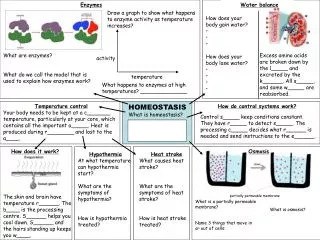
Processes used to maintain homeostasis: 1. Diffusion: net movement of particles from an area of higher concentration to an area of lower concentration until it reaches equilibrium *results due to the random movement of particles, a slow process
3 Key factors of diffusion 1. Concentration: more concentrated, more rapid diffusion 2. Temperature: increase in temp. speeds up the movement of molecules and then diffusion 3. Pressure: increase in pressure, accelerate molecule movement and diffusion
Goal of cell is to have: • Dynamic Equilibrium: continuous movement of molecules, same rate in and out of the cell, but no overall concentration change • Concentration gradient: difference in concentration across space
Processes used to maintain homeostasis continued… 2. Osmosis: diffusion of water across a semi-permeable membrane • water flows to side of cell with less water, where water concentration is low • *water continues to diffuse until it is in equalconcentration on both sides of the membrane
3 types of solutions, osmosis in cells 1. Isotonic solution: The concentration of solute inside and outside the cell are the same, so water moves in and out of the cell at the same rate • *shape and size of the cell are normal, do not experience osmosis, water concentration is the same in and out of the cell
3 types of solutions, osmosis in cells 2. Hypotonic: solute concentration is low outside the cell, water concentration or amount is lower inside the cell so water moves into the cell • *shape and size of cell swells beyond normal pressure increases, can cause cytolysis • Result: animal cells can burst, plant cells become firm due to turgor pressure
3 types of solutions, osmosis in cells 3. Hypertonic: solute concentration outside the cell is higher than in the cell, so water concentration or amount is less outside the cell, so water moves out of the cell • *size of cell decrease for animal cells, causes plasmolysis Result: animal cell shrinks, vacuole in plant cell shrinks, plant wilts
Turgor pressure vs. plasmolysis vs. cytolysis • Turgor pressure: in hypotonic cells, when water moves into cell the pressure increases and makes cell rigid • Plasmolysis: water leaves cell and cell wilts • Cytolysis: cells take in too much water and burst, only in animal cells
Water regulation in organisms • Single celled: most found in hypotonic environments, lakes and ponds *Want to avoid bursting, have contractile vacuoles that act as a pump and remove water by contracting
2. Higher level animals: cells in a hypertonic environment: no water, high-salt • Small portion of cells in the brain shrink and cause thirst • Cells in hypotonic environment drink too much water, low salt • *Small patch of cells in brain when they swell trigger nerves in kidney and cause kidney tubules to open up and produce more urine
Passive transport: no energy is needed to move the particles across the membrane • Ex. Diffusion and osmosis
Facilitated diffusion: passive transport of materials with the help of proteins • Moves sugars and amino acids