Understanding Voltaic Cells: An Exciting Chemistry Experiment
Learn about the components of a voltaic cell - anode, cathode, and salt bridge - and conduct an engaging lab to witness electrochemical reactions in action. Dive into the world of chemistry with this hands-on activity!

Understanding Voltaic Cells: An Exciting Chemistry Experiment
E N D
Presentation Transcript
Voltaic Cell Lab Chemistry in the Community A
Warm Up • What are the different parts to a voltaic cell?
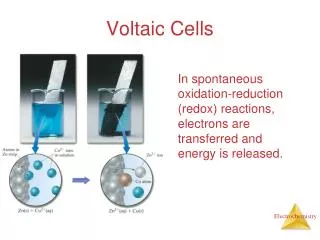
Answer • Anode-Where oxidation takes place • Cathode-Where reduction takes place (RED CAT*) • Salt Bridge- keeps solutions electrically neutral and allows the free flow of ions from one cell to another.
I can…… • I can demonstrate how a voltaic cell works.
Activities • Pre-lab • Lab
Pre-Lab • Draw a voltaic cell and label and explain the parts of the cell • Read through the lab and come up with a hypothesis
materials • Liquids • Copper Nitrate • Zinc Nitrate • Magnesium Nitrate • Iron Nitrate • Metals • Copper • Zinc • Magnesium • Iron
Lab Start lab in groups of 3-4
Cool Down • What does a salt bridge do?